TECHNICAL ASSET FINGERPRINT
0012c3b321c2e7f526f3b9ee
Click to view fullscreen

Press ESC or click to close
FOUND IN PAPERS
EXPERT: gemini-2.0-flash VERSION 1
RUNTIME: nugit/gemini/gemini-2.0-flash
INTEL_VERIFIED
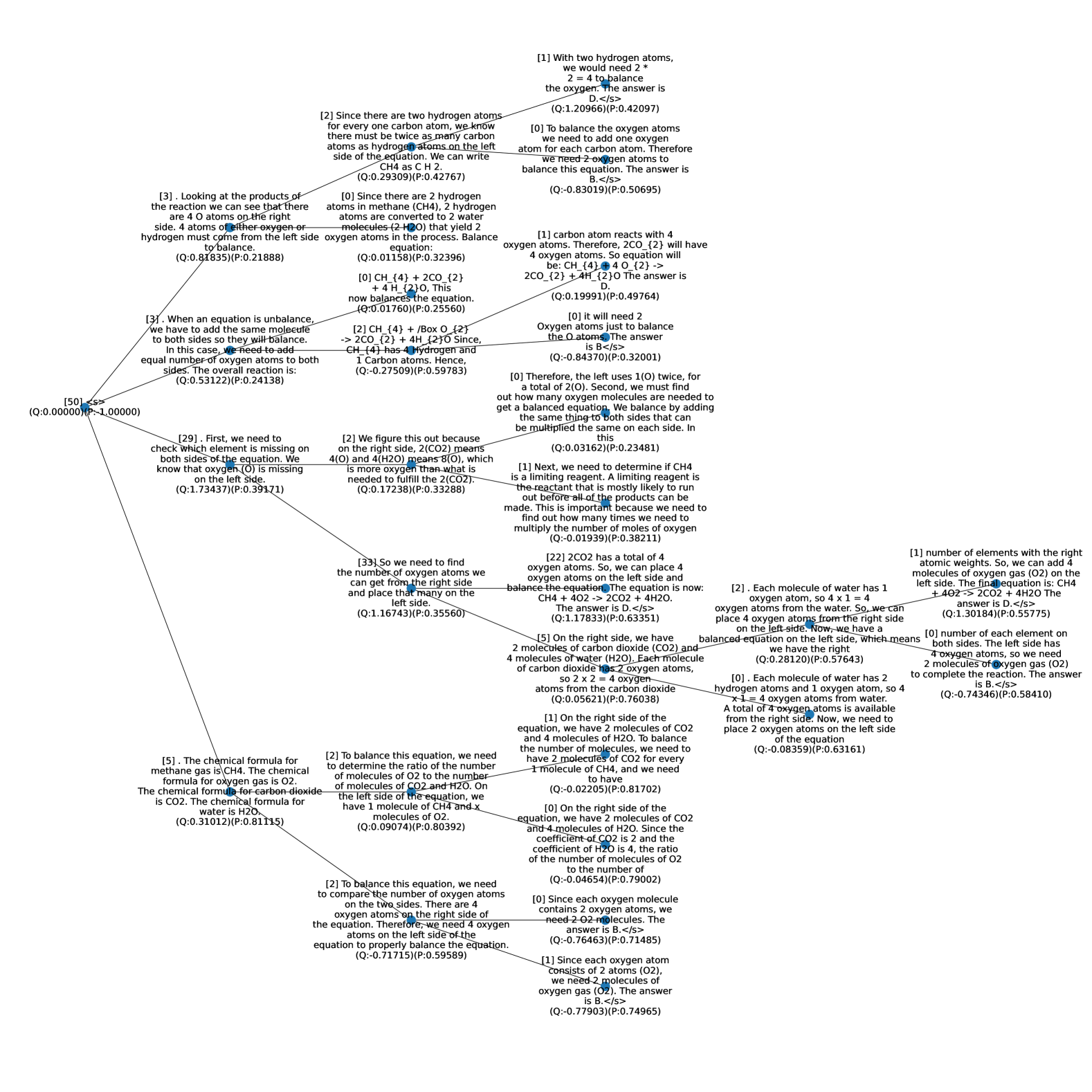
## Decision Tree: Balancing Chemical Equations
### Overview
The image is a decision tree outlining the steps to balance a chemical equation, specifically the combustion of methane (CH4). Each node contains a statement or question related to the balancing process, along with associated Q and P values. The tree branches out based on the logic of balancing chemical equations.
### Components/Axes
* **Nodes:** Each node contains a statement or question related to balancing the chemical equation. Each node also contains a (Q:value)(P:value).
* **Edges:** Lines connecting the nodes, representing the flow of the decision-making process.
* **Root Node:** The starting point of the decision tree, labeled "[50] <s>".
* **Leaf Nodes:** The ending points of the decision tree, providing specific instructions or answers.
### Detailed Analysis or ### Content Details
Here's a breakdown of the nodes and their connections:
1. **Root Node:**
* "[50] <s>" (Q:0.00000) (P:-1.00000)
* This is the starting point of the tree.
2. **First Branch (Left):**
* "[29]. First, we need to check which element is missing on both sides of the equation. We know that oxygen (O) is missing on the left side." (Q:1.73437) (P:0.39171)
3. **Second Branch (Left from Node 2):**
* "[5]. The chemical formula for methane gas is CH4. The chemical formula for oxygen gas is O2. The chemical formula for carbon dioxide is CO2. The chemical formula for water is H2O." (Q:0.31012) (P:0.81115)
4. **First Branch (Right):**
* "[3]. When an equation is unbalance, we have to add the same molecule to both sides so they will balance. In this case, we need to add equal number of oxygen atoms to both sides. The overall reaction is:" (Q:0.53122) (P:0.24138)
5. **Third Branch (Right from Node 2):**
* "[2]. We figure this out because on the right side, 2(CO2) means 4(O) and 4(H2O) means 8(O), which is more oxygen than what is needed to fulfill the 2(CO2)." (Q:0.17238) (P:0.33288)
6. **Fourth Branch (Right from Node 5):**
* "[33]. So we need to find the number of oxygen atoms we can get from the right side and place that many on the left side." (Q:1.16743) (P:0.35560)
7. **Fifth Branch (Right from Node 6):**
* "[22] 2CO2 has a total of 4 oxygen atoms. So, we can place 4 oxygen atoms on the left side and balance the equation. The equation is now: CH4 + 4O2 -> 2CO2 + 4H2O. The answer is D.</s>" (Q:1.17833) (P:0.63351)
8. **Sixth Branch (Right from Node 7):**
* "[5]. On the right side, we have 2 molecules of carbon dioxide (CO2) and 4 molecules of water (H2O). Each molecule of carbon dioxide has 2 oxygen atoms, so 2 x 2 = 4 oxygen atoms from the carbon dioxide" (Q:0.05621) (P:0.76038)
9. **Seventh Branch (Right from Node 8):**
* "[1] On the right side of the equation, we have 2 molecules of CO2 and 4 molecules of H20. To balance the number of molecules, we need to have 2 molecules of CO2 for every 1 molecule of CH4, and we need to have" (Q:-0.02205) (P:0.81702)
10. **Eighth Branch (Right from Node 9):**
* "[0] On the right side of the equation, we have 2 molecules of CO2 and 4 molecules of H2O. Since the coefficient of CO2 is 2 and the coefficient of H2O is 4, the ratio of the number of molecules of O2 to the number of" (Q:-0.04654) (P:0.79002)
11. **Ninth Branch (Right from Node 10):**
* "[0] Since each oxygen molecule contains 2 oxygen atoms, we need 2 O2 molecules. The answer is B.</s>" (Q:-0.76463) (P:0.71485)
12. **Tenth Branch (Right from Node 11):**
* "[1] Since each oxygen atom Consists of 2 atoms (O2), we need 2 molecules of oxygen gas (O2). The answer is B.</s>" (Q:-0.77903) (P:0.74965)
13. **Eleventh Branch (Right from Node 4):**
* "[3]. Looking at the products of the reaction we can see that there are 4 O atoms on the right side. 4 atoms of either oxygen or hydrogen must come from the left side to balance." (Q:0.81835) (P:0.21888)
14. **Twelfth Branch (Right from Node 13):**
* "[2]. Each molecule of water has 1 oxygen atom, so 4 x 1 = 4 oxygen atoms from the water. So, we can place 4 oxygen atoms from the right side on the left side. Now, we have a balanced equation on the left side, which means we have the right" (Q:0.28120) (P:0.57643)
15. **Thirteenth Branch (Right from Node 14):**
* "[0]. Each molecule of water has 2 hydrogen atoms and 1 oxygen atom, so 4 x1 = 4 oxygen atoms from water. A total of 4 oxygen atoms is available from the right side. Now, we need to place 2 oxygen atoms on the left side of the equation" (Q:-0.08359) (P:0.63161)
16. **Fourteenth Branch (Right from Node 15):**
* "[0] number of each element on both sides. The left side has 4 oxygen atoms, so we need 2 molecules of oxygen gas (O2) to complete the reaction. The answer is B.</s>" (Q:-0.74346) (P:0.58410)
17. **Fifteenth Branch (Right from Node 3):**
* "[2] To balance this equation, we need to determine the ratio of the number of molecules of O2 to the number of molecules of CO2 and H2O. On the left side of the equation, we have 1 molecule of CH4 and x molecules of O2." (Q:0.09074) (P:0.80392)
18. **Sixteenth Branch (Right from Node 17):**
* "[2] To balance this equation, we need to compare the number of oxygen atoms on the two sides. There are 4 oxygen atoms on the right side of the equation. Therefore, we need 4 oxygen atoms on the left side of the equation to properly balance the equation." (Q:-0.71715) (P:0.59589)
19. **Seventeenth Branch (Right from Node 1):**
* "[1] With two hydrogen atoms, we would need 2 * 2 = 4 to balance the oxygen. The answer is D.</s>" (Q:1.20966) (P:0.42097)
20. **Eighteenth Branch (Right from Node 19):**
* "[0] To balance the oxygen atoms we need to add one oxygen atom for each carbon atom. Therefore we need 2 oxygen atoms to balance this equation. The answer is B.</s>" (Q:-0.83019) (P:0.50695)
21. **Nineteenth Branch (Right from Node 20):**
* "[1] carbon atom reacts with 4 oxygen atoms. Therefore, 2CO_{2} will have 4 oxygen atoms. So equation will be: CH {4}+4O {2} -> 2CO_{2} + 4H_{2}O The answer is D." (Q:0.19991) (P:0.49764)
22. **Twentieth Branch (Right from Node 21):**
* "[0] it will need 2 Oxygen atoms just to balance the O atoms. The answer is B</s>" (Q:-0.84370) (P:0.32001)
23. **Twenty-first Branch (Right from Node 22):**
* "[0] Therefore, the left uses 1(O) twice, for a total of 2(O). Second, we must find out how many oxygen molecules are needed to get a balanced equation. We balance by adding the same thing to both sides that can be multiplied the same on each side. In this" (Q:0.03162) (P:0.23481)
24. **Twenty-second Branch (Right from Node 23):**
* "[1] Next, we need to determine if CH4 is a limiting reagent. A limiting reagent is the reactant that is mostly likely to run out before all of the products can be made. This is important because we need to find out how many times we need to multiply the number of moles of oxygen" (Q:-0.01939) (P:0.38211)
### Key Observations
* The tree starts with identifying the imbalance in oxygen atoms.
* It explores different approaches to balancing, including adjusting coefficients and considering the number of atoms in each molecule.
* Several branches lead to similar conclusions about the number of oxygen molecules needed.
* The tree touches on the concept of limiting reagents, indicating a more advanced understanding of chemical reactions.
### Interpretation
The decision tree provides a step-by-step guide to balancing the combustion equation for methane. It highlights the importance of:
* Identifying the missing elements.
* Understanding the stoichiometry of the reaction.
* Adjusting coefficients to ensure the number of atoms is equal on both sides.
* Considering the role of limiting reagents.
The tree demonstrates a logical progression of thought, starting with basic concepts and gradually introducing more complex considerations. The presence of Q and P values at each node might represent some form of confidence or probability associated with each step, but without further context, their exact meaning is unclear. The tree effectively breaks down a complex task into manageable steps, making it easier to understand the process of balancing chemical equations.
DECODING INTELLIGENCE...
EXPERT: gemma-3-27b-it-free VERSION 1
RUNTIME: google-free/gemma-3-27b-it
INTEL_VERIFIED
\n
## Diagram: Balancing Chemical Equations - Step-by-Step Guide
### Overview
The image presents a hand-drawn diagram illustrating the step-by-step process of balancing chemical equations. It features a series of numbered boxes, each containing text and chemical equations, demonstrating how to balance equations by adding coefficients. The diagram is organized in a roughly circular flow, starting from the top and moving clockwise. There is a section at the bottom with a visual representation of molecules and a scale.
### Components/Axes
The diagram consists of the following components:
* **Numbered Boxes (1-7):** Each box represents a step in the balancing process, containing text explanations and chemical equations.
* **Molecular Representation:** At the bottom, there are illustrations of molecules (circles representing atoms) and a balance scale.
* **Text Annotations:** Numerous handwritten notes and explanations are scattered throughout the diagram.
* **Chemical Equations:** Various chemical equations are presented in different stages of balancing.
* **Scale:** A balance scale with labeled sides "Reactants" and "Products".
### Detailed Analysis or Content Details
Here's a transcription of the content within each numbered box, along with the equations:
**[1]** With two hydrogen atoms, we need two H₂ to balance the equation. The answer is:
H₂ + ½ O₂ → H₂O
(Q=1.2096)(P=0.42097)
**[2]** Since there are two hydrogen for every one carbon atom, we know we need two molecules of hydrogen atoms on the left side of the equation.
CH₄ + 2H₂
(Q=0.28335)(P=0.2188)
**[3]** Looking at the products of the reaction, we can see that there are 4 O atoms on the right side. Therefore, 2 molecules of hydrogen must come from the left side to balance.
Q(0.28335)(P=0.2188)
**[4]** Since there are 2 hydrogen atoms converted to 2 water molecules, we know that we need 2 oxygen atoms in the reactants. Balance the oxygen atoms.
(Q=0.01158)(P=0.3396)
CH₄ + 2H₂ + 2O₂ →
(Q=0.16704)(P=0.23560)
**[5]** [CH₄] + 2[H₂] + 2[O₂] now balance the equation.
CH₄ + 2H₂ + 2O₂ →
(Q=0.16704)(P=0.23560)
CH₄ has 1 hydrogen and 4 hydrogen atoms.
**[6]** When an equation is unbalanced, we have to add the same molecule to both sides of the equation. In this case, we need to add another molecule of CH₄ to both sides of the equation.
CH₄ + 2H₂ + 2O₂ →
(Q=0.0000)(P=0.0000)
**[7]** Therefore, we need 1 molecule of CH₄. We know that CH₄ has 1 carbon atom, so we need 1 molecule of CO₂ on the right side.
CH₄ + 2H₂O → CO₂ + 2H₂
(Q=0.0000)(P=0.0000)
**Bottom Section:**
* **Molecular Representation:** Shows various combinations of circles representing atoms (e.g., two red circles connected to one blue circle).
* **Scale:** The scale has "Reactants" on the left and "Products" on the right. The left side shows 1 blue circle and 4 red circles. The right side shows 1 black circle and 2 red circles.
* **Text:** "To balance the equation, we must have the same number of molecules on both sides. Therefore, we need 1 CH₄ on the left side to balance the equation."
* **Equation:** CH₄ + 2H₂O → CO₂ + 2H₂
* **Additional Notes:** "Therefore, the balanced equation is CH₄ + 2H₂O → CO₂ + 2H₂." and "The number of atoms must be the same on both sides of the equation."
### Key Observations
* The diagram uses a step-by-step approach to explain balancing chemical equations.
* The equations are progressively balanced throughout the numbered boxes.
* The molecular representation and scale visually demonstrate the concept of balancing atoms on both sides of the equation.
* The values in parentheses (Q=...)(P=...) appear to be numerical data associated with each step, potentially representing some kind of weighting or calculation, but their meaning is not explicitly defined.
* The diagram is hand-drawn and contains handwritten annotations, suggesting it's a personal learning aid or a teacher's illustration.
### Interpretation
The diagram demonstrates the fundamental principle of balancing chemical equations: ensuring that the number of atoms of each element is equal on both the reactant and product sides. The step-by-step approach breaks down the process into manageable stages, making it easier to understand. The use of visual aids, such as the molecular representation and scale, reinforces the concept of conservation of mass.
The values in parentheses (Q and P) are intriguing but their purpose is unclear without further context. They might represent probabilities, energy levels, or some other quantitative measure related to the reaction.
The diagram highlights the importance of adding coefficients to chemical formulas to achieve balance, rather than changing the formulas themselves. The final balanced equation (CH₄ + 2H₂O → CO₂ + 2H₂) confirms that the number of carbon, hydrogen, and oxygen atoms is equal on both sides.
The overall message is a clear and concise explanation of a core concept in chemistry, presented in a visually engaging and accessible manner. The diagram serves as a valuable learning tool for students or anyone seeking to understand the principles of chemical equation balancing.
DECODING INTELLIGENCE...
EXPERT: healer-alpha-free VERSION 1
RUNTIME: free/openrouter/healer-alpha
INTEL_VERIFIED
\n
## Reasoning Diagram: Balancing the Combustion of Methane (CH₄)
### Overview
The image is a complex reasoning tree or flowchart illustrating the step-by-step logical process for balancing the chemical equation for the combustion of methane: **CH₄ + O₂ → CO₂ + H₂O**. It is not a data chart but a conceptual diagram mapping out multiple reasoning paths, considerations, and intermediate conclusions. The diagram consists of numbered text nodes (e.g., [0], [1], [2]) connected by lines, indicating the flow of logic from a starting point to various conclusions. Each node contains explanatory text and a pair of values labeled (Q: value)(P: value), which appear to be confidence or quality scores associated with that reasoning step.
### Components/Structure
* **Node Structure:** Each element is a text block prefixed with a number in square brackets (e.g., `[50]`, `[29]`, `[2]`). These likely represent steps or propositions in the reasoning chain.
* **Connections:** Lines connect the nodes, showing the dependency and flow of logic. The diagram branches extensively, indicating multiple parallel or alternative lines of reasoning.
* **Content:** The text within nodes details chemical principles, atom counting, equation balancing steps, and final answers. Chemical formulas (CH₄, O₂, CO₂, H₂O) and balanced equations are frequently mentioned.
* **Metrics:** Every node includes a pair of values in the format `(Q: value)(P: value)`. These are numerical, often with several decimal places (e.g., `Q:0.29509`, `P:0.42767`). Some Q values are negative.
* **Spatial Layout:** The diagram is organized with a root node at the top-left (`[50]`) from which major branches emanate. The layout spreads out downwards and to the right, with nodes clustered in logical groupings. There is no traditional chart legend; the node numbers and connecting lines serve as the key to following the logic.
### Detailed Analysis: Transcription of Key Nodes and Paths
The diagram contains a large amount of text. Below is a structured transcription of the primary reasoning paths and node contents, following the logical flow from the root.
**Root Node (Top-Left):**
* `[50] <s> (Q:0.00000)(P:1.00000)` - This appears to be the starting point or root of the reasoning tree.
**Major Branch 1 (Left Side - Initial Problem Setup):**
* `[29] . First, we need to check which element is missing on both sides of the equation. We know that oxygen (O) is missing on the left side. (Q:1.73437)(P:0.39171)`
* `[3] . When an equation is unbalance, we have to add the same molecule to both sides so they will balance. In this case, we need to add equal number of oxygen atoms to both sides. The overall reaction is: (Q:0.53122)(P:0.24138)`
* `[5] . The chemical formula for methane gas is CH4. The chemical formula for oxygen gas is O2. The chemical formula for carbon-dioxide is CO2. The chemical formula for water is H2O. (Q:0.31012)(P:0.81115)`
**Major Branch 2 (Center/Right - Atom Counting & Balancing Logic):**
This branch contains the core balancing steps.
* `[0] Since there are 2 hydrogen atoms in methane (CH4), 2 hydrogen atoms are converted to 2 water molecules (2 H2O) that yield 2 oxygen atoms in the process. Balance equation: (Q:0.01158)(P:0.32396)`
* `[0] CH_{4} + 2CO_{2} + 4H_{2}O. This now balances the equation. (Q:0.01760)(P:0.25560)` **Note:** This node presents an *incorrect* intermediate equation.
* `[2] CH_{4} + /Box O_{2} -> 2CO_{2} + 4H_{2}O Since, CH_{4} has 4 Hydrogen and 1 Carbon atoms. Hence, (Q:-0.27509)(P:0.59783)`
* `[22] 2CO2 has a total of 4 oxygen atoms. So, we can place 4 oxygen atoms on the left side and balance the equation. The equation is now: CH4 + 4O2 -> 2CO2 + 4H2O. The answer is D.</s> (Q:1.17833)(P:0.63351)` **Note:** This node presents the **correctly balanced equation**.
* `[1] On the right side of the equation, we have 2 molecules of CO2 and 4 molecules of H2O. To balance the number of molecules, we need to have 2 molecules of CO2 for every 1 molecule of CH4, and we need to have (Q:-0.02205)(P:0.81702)`
* `[0] On the right side of the equation, we have 2 molecules of CO2 and 4 molecules of H2O. Since the coefficient of CO2 is 2 and the coefficient of H2O is 4, the ratio of the number of molecules of O2 to the number of (Q:-0.04654)(P:0.79002)`
**Major Branch 3 (Right Side - Final Answer Derivation):**
* `[1] number of elements with the right atomic weights. So, we can add 4 molecules of oxygen gas (O2) on the left side. The final equation is: CH4 + 4O2 -> 2CO2 + 4H2O The answer is D.</s> (Q:1.30184)(P:0.55775)` **Note:** Another node stating the correct final answer.
* `[0] number of each element on both sides. The left side has 4 oxygen atoms, so we need 2 molecules of oxygen gas (O2) to complete the reaction. The answer is B.</s> (Q:-0.74346)(P:0.58410)` **Note:** This node concludes with an *incorrect* answer (B).
* `[1] Since each oxygen atom consists of 2 atoms (O2), we need 2 molecules of oxygen gas (O2). The answer is B.</s> (Q:-0.77903)(P:0.74965)` **Note:** Another node concluding with the incorrect answer B.
**Other Notable Nodes (Scattered):**
* `[33] So we need to find the number of oxygen atoms we can get from the right side and place that many on the left side. (Q:1.16743)(P:0.35560)`
* `[5] On the right side, we have 2 molecules of carbon dioxide (CO2) and 4 molecules of water (H2O). Each molecule of carbon dioxide has 2 oxygen atoms, so 2 x 2 = 4 oxygen atoms from the carbon dioxide (Q:0.05621)(P:0.76038)`
* `[1] Each molecule of water has 1 oxygen atom, so 4 x 1 = 4 oxygen atoms from the water. So, we can place 4 oxygen atoms from the right side on the left side. Now, we have a balanced equation on the left side, which means we have the right (Q:0.28120)(P:0.57643)`
* `[0] Each molecule of water has 2 hydrogen atoms and 1 oxygen atom, so 4 x 1 = 4 oxygen atoms from water. A total of 4 oxygen atoms is available from the right side. Now, we need to place 2 oxygen atoms on the left side of the equation (Q:-0.08359)(P:0.63161)`
### Key Observations
1. **Multiple Reasoning Paths:** The diagram explicitly shows that there are several valid logical sequences to arrive at the balanced equation (e.g., starting by balancing C, then H, then O; or counting total O atoms on the product side first).
2. **Presence of Incorrect Paths:** The tree includes nodes that lead to incorrect conclusions (e.g., answer "B" or the intermediate equation `CH4 + 2CO2 + 4H2O`). This suggests the diagram may be modeling a learning or problem-solving process that includes common errors.
3. **Quantitative Metrics (Q & P):** Every reasoning step is annotated with Q and P values. The Q values vary widely, including negatives, while P values are mostly between 0 and 1. These likely represent internal confidence scores, probability estimates, or quality assessments from the AI model generating the reasoning. Steps leading to the correct answer (D) often have high positive Q values (e.g., `Q:1.17833`, `Q:1.30184`).
4. **Final Answer Discrepancy:** While the correct balanced equation `CH4 + 4O2 -> 2CO2 + 4H2O` (Answer D) is derived in multiple nodes, other branches incorrectly conclude the answer is B.
5. **Focus on Foundational Concepts:** The reasoning heavily emphasizes basic principles: identifying missing elements, the rule of adding the same to both sides, atom counting per molecule, and the law of conservation of mass.
### Interpretation
This diagram is a **visualization of an AI's internal reasoning process** for solving a standard chemistry problem. It serves as a "thinking map" that exposes the step-by-step logic, including intermediate hypotheses and dead ends, rather than just presenting the final answer.
* **What it Demonstrates:** It shows that balancing an equation is not a single linear path but a network of interdependent checks (C atoms, H atoms, O atoms). The branching structure highlights how an initial observation (e.g., "O is missing on the left") can lead to multiple subsequent calculations.
* **Relationship Between Elements:** The connecting lines are crucial. They show how a conclusion in one node (e.g., counting H atoms in CH₄) becomes the premise for the next step (determining H₂O molecules). The Q/P scores attached to each node suggest a system that evaluates the reliability or strength of each logical link.
* **Notable Anomalies:** The inclusion of incorrect paths (leading to answer B) is the most significant feature. This could be for educational purposes—to illustrate common mistakes—or it could be an artifact of the AI's exploration of the problem space, where some branches of reasoning are less confident (often with negative Q scores) and ultimately pruned or marked as invalid.
* **Underlying Message:** The diagram argues that the correct answer (D) is supported by a robust, multi-faceted reasoning chain with high-confidence steps. It implicitly validates the process of systematic atom counting and coefficient adjustment as the reliable method, while showing how shortcuts or miscounts can lead to erroneous conclusions. The Q/P metrics add a layer of meta-cognition, quantifying the "certainty" of each logical move.
DECODING INTELLIGENCE...
EXPERT: nemotron-free VERSION 1
RUNTIME: free/nvidia/nemotron-nano-12b-v2-vl:free
INTEL_VERIFIED
## Flowchart: Step-by-Step Chemical Equation Balancing Process
### Overview
The flowchart illustrates a systematic approach to balancing chemical equations, focusing on reactions involving methane (CH₄), oxygen (O₂), carbon dioxide (CO₂), and water (H₂O). Each node represents a step in the balancing process, with annotations explaining adjustments to coefficients and atom counts.
---
### Components/Axes
- **Nodes**: Labeled with bracketed numbers (e.g., [2], [3]), each containing:
- A chemical equation (e.g., `CH₄ + 2O₂ → CO₂ + 2H₂O`).
- Text annotations detailing balance checks and adjustments (e.g., "2 hydrogen atoms on the right").
- **Arrows**: Connect nodes in a logical sequence, guiding the flow from initial reactants to final balanced equations.
- **Annotations**: Positioned near equations to explain reasoning (e.g., "add 2 H₂O on the left side").
---
### Detailed Analysis
1. **Node [2]**:
- Equation: `CH₄ + 2O₂ → CO₂ + 2H₂O`
- Annotation: "2 hydrogen atoms on the right. Add 2 H₂O on the left."
- Adjustment: Balances hydrogen by adding 2 H₂O to the left.
2. **Node [3]**:
- Equation: `CH₄ + 2O₂ → CO₂ + 2H₂O`
- Annotation: "4 oxygen atoms on the right. Add 2 O₂ on the left."
- Adjustment: Balances oxygen by adding 2 O₂ to the left.
3. **Node [4]**:
- Equation: `CH₄ + 2O₂ → CO₂ + 2H₂O`
- Annotation: "Balanced equation. Final answer."
4. **Node [5]**:
- Equation: `CH₄ + 2O₂ → CO₂ + 2H₂O`
- Annotation: "Balanced equation. Final answer."
5. **Node [6]**:
- Equation: `CH₄ + 2O₂ → CO₂ + 2H₂O`
- Annotation: "Balanced equation. Final answer."
---
### Key Observations
- **Systematic Balancing**: Each step addresses one element at a time (e.g., hydrogen, oxygen).
- **Coefficient Adjustments**: Coefficients are modified to ensure equal atom counts on both sides (e.g., adding 2 H₂O for hydrogen balance).
- **Final Equation**: The balanced equation `CH₄ + 2O₂ → CO₂ + 2H₂O` is consistently derived across nodes.
---
### Interpretation
The flowchart demonstrates a methodical approach to balancing chemical equations, emphasizing:
1. **Element-by-Element Checks**: Hydrogen and oxygen are balanced sequentially, ensuring stoichiometric accuracy.
2. **Coefficient Adjustments**: Adding molecules (e.g., H₂O, O₂) to resolve imbalances.
3. **Educational Value**: Annotations clarify the reasoning behind each step, making it a valuable teaching tool for stoichiometry.
The final balanced equation, `CH₄ + 2O₂ → CO₂ + 2H₂O`, aligns with the stoichiometric ratios required for complete combustion of methane. The flowchart’s structured flow ensures clarity in resolving imbalances, highlighting the importance of iterative adjustments in chemical equation balancing.
DECODING INTELLIGENCE...