TECHNICAL ASSET FINGERPRINT
0012c3b321c2e7f526f3b9ee
Click to view fullscreen

Press ESC or click to close
FOUND IN PAPERS
EXPERT: gemini-2.0-flash VERSION 1
RUNTIME: nugit/gemini/gemini-2.0-flash
INTEL_VERIFIED
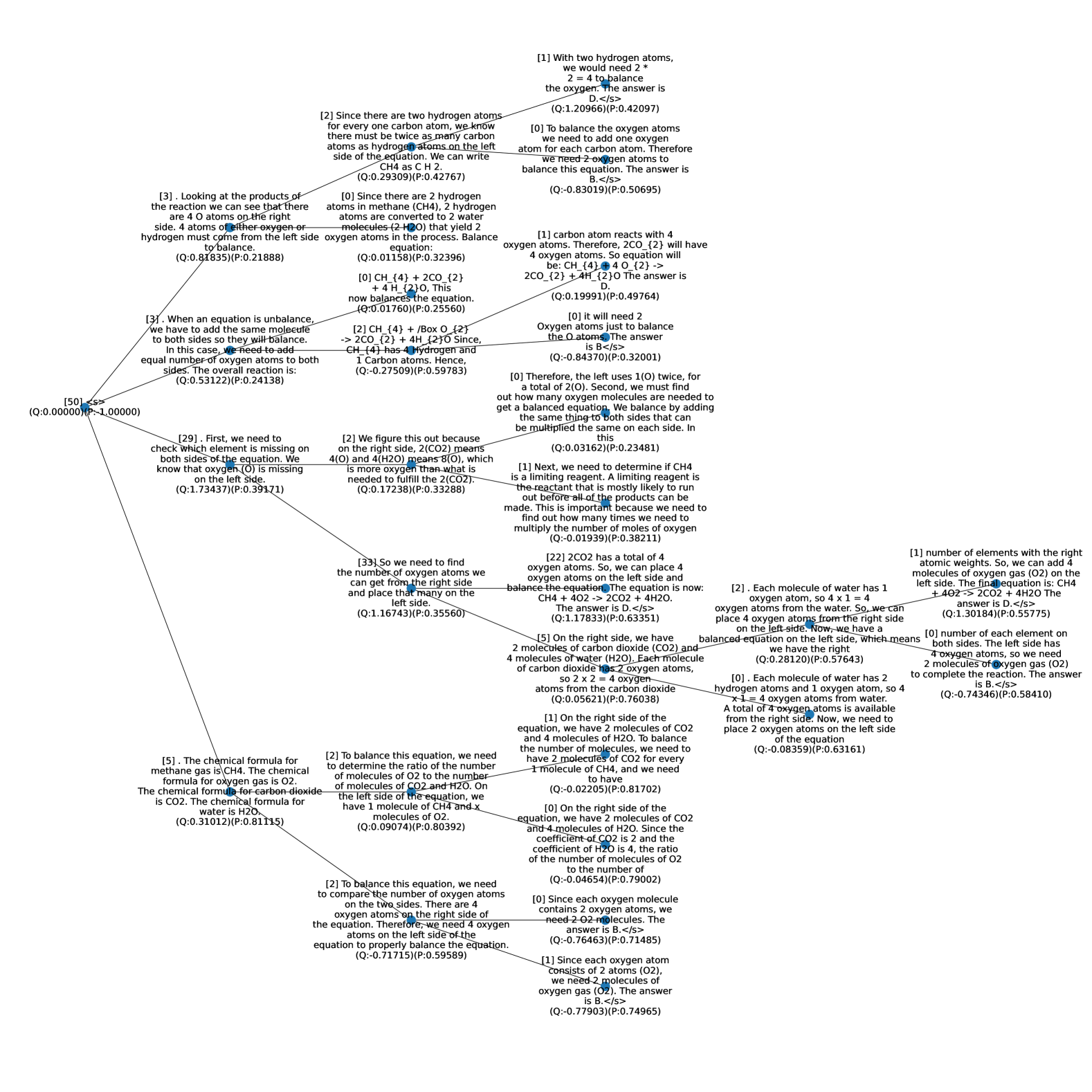
## Decision Tree: Balancing Chemical Equations
### Overview
The image is a decision tree outlining the steps to balance a chemical equation, specifically the combustion of methane (CH4). Each node contains a statement or question related to the balancing process, along with associated Q and P values. The tree branches out based on the logic of balancing chemical equations.
### Components/Axes
* **Nodes:** Each node contains a statement or question related to balancing the chemical equation. Each node also contains a (Q:value)(P:value).
* **Edges:** Lines connecting the nodes, representing the flow of the decision-making process.
* **Root Node:** The starting point of the decision tree, labeled "[50] <s>".
* **Leaf Nodes:** The ending points of the decision tree, providing specific instructions or answers.
### Detailed Analysis or ### Content Details
Here's a breakdown of the nodes and their connections:
1. **Root Node:**
* "[50] <s>" (Q:0.00000) (P:-1.00000)
* This is the starting point of the tree.
2. **First Branch (Left):**
* "[29]. First, we need to check which element is missing on both sides of the equation. We know that oxygen (O) is missing on the left side." (Q:1.73437) (P:0.39171)
3. **Second Branch (Left from Node 2):**
* "[5]. The chemical formula for methane gas is CH4. The chemical formula for oxygen gas is O2. The chemical formula for carbon dioxide is CO2. The chemical formula for water is H2O." (Q:0.31012) (P:0.81115)
4. **First Branch (Right):**
* "[3]. When an equation is unbalance, we have to add the same molecule to both sides so they will balance. In this case, we need to add equal number of oxygen atoms to both sides. The overall reaction is:" (Q:0.53122) (P:0.24138)
5. **Third Branch (Right from Node 2):**
* "[2]. We figure this out because on the right side, 2(CO2) means 4(O) and 4(H2O) means 8(O), which is more oxygen than what is needed to fulfill the 2(CO2)." (Q:0.17238) (P:0.33288)
6. **Fourth Branch (Right from Node 5):**
* "[33]. So we need to find the number of oxygen atoms we can get from the right side and place that many on the left side." (Q:1.16743) (P:0.35560)
7. **Fifth Branch (Right from Node 6):**
* "[22] 2CO2 has a total of 4 oxygen atoms. So, we can place 4 oxygen atoms on the left side and balance the equation. The equation is now: CH4 + 4O2 -> 2CO2 + 4H2O. The answer is D.</s>" (Q:1.17833) (P:0.63351)
8. **Sixth Branch (Right from Node 7):**
* "[5]. On the right side, we have 2 molecules of carbon dioxide (CO2) and 4 molecules of water (H2O). Each molecule of carbon dioxide has 2 oxygen atoms, so 2 x 2 = 4 oxygen atoms from the carbon dioxide" (Q:0.05621) (P:0.76038)
9. **Seventh Branch (Right from Node 8):**
* "[1] On the right side of the equation, we have 2 molecules of CO2 and 4 molecules of H20. To balance the number of molecules, we need to have 2 molecules of CO2 for every 1 molecule of CH4, and we need to have" (Q:-0.02205) (P:0.81702)
10. **Eighth Branch (Right from Node 9):**
* "[0] On the right side of the equation, we have 2 molecules of CO2 and 4 molecules of H2O. Since the coefficient of CO2 is 2 and the coefficient of H2O is 4, the ratio of the number of molecules of O2 to the number of" (Q:-0.04654) (P:0.79002)
11. **Ninth Branch (Right from Node 10):**
* "[0] Since each oxygen molecule contains 2 oxygen atoms, we need 2 O2 molecules. The answer is B.</s>" (Q:-0.76463) (P:0.71485)
12. **Tenth Branch (Right from Node 11):**
* "[1] Since each oxygen atom Consists of 2 atoms (O2), we need 2 molecules of oxygen gas (O2). The answer is B.</s>" (Q:-0.77903) (P:0.74965)
13. **Eleventh Branch (Right from Node 4):**
* "[3]. Looking at the products of the reaction we can see that there are 4 O atoms on the right side. 4 atoms of either oxygen or hydrogen must come from the left side to balance." (Q:0.81835) (P:0.21888)
14. **Twelfth Branch (Right from Node 13):**
* "[2]. Each molecule of water has 1 oxygen atom, so 4 x 1 = 4 oxygen atoms from the water. So, we can place 4 oxygen atoms from the right side on the left side. Now, we have a balanced equation on the left side, which means we have the right" (Q:0.28120) (P:0.57643)
15. **Thirteenth Branch (Right from Node 14):**
* "[0]. Each molecule of water has 2 hydrogen atoms and 1 oxygen atom, so 4 x1 = 4 oxygen atoms from water. A total of 4 oxygen atoms is available from the right side. Now, we need to place 2 oxygen atoms on the left side of the equation" (Q:-0.08359) (P:0.63161)
16. **Fourteenth Branch (Right from Node 15):**
* "[0] number of each element on both sides. The left side has 4 oxygen atoms, so we need 2 molecules of oxygen gas (O2) to complete the reaction. The answer is B.</s>" (Q:-0.74346) (P:0.58410)
17. **Fifteenth Branch (Right from Node 3):**
* "[2] To balance this equation, we need to determine the ratio of the number of molecules of O2 to the number of molecules of CO2 and H2O. On the left side of the equation, we have 1 molecule of CH4 and x molecules of O2." (Q:0.09074) (P:0.80392)
18. **Sixteenth Branch (Right from Node 17):**
* "[2] To balance this equation, we need to compare the number of oxygen atoms on the two sides. There are 4 oxygen atoms on the right side of the equation. Therefore, we need 4 oxygen atoms on the left side of the equation to properly balance the equation." (Q:-0.71715) (P:0.59589)
19. **Seventeenth Branch (Right from Node 1):**
* "[1] With two hydrogen atoms, we would need 2 * 2 = 4 to balance the oxygen. The answer is D.</s>" (Q:1.20966) (P:0.42097)
20. **Eighteenth Branch (Right from Node 19):**
* "[0] To balance the oxygen atoms we need to add one oxygen atom for each carbon atom. Therefore we need 2 oxygen atoms to balance this equation. The answer is B.</s>" (Q:-0.83019) (P:0.50695)
21. **Nineteenth Branch (Right from Node 20):**
* "[1] carbon atom reacts with 4 oxygen atoms. Therefore, 2CO_{2} will have 4 oxygen atoms. So equation will be: CH {4}+4O {2} -> 2CO_{2} + 4H_{2}O The answer is D." (Q:0.19991) (P:0.49764)
22. **Twentieth Branch (Right from Node 21):**
* "[0] it will need 2 Oxygen atoms just to balance the O atoms. The answer is B</s>" (Q:-0.84370) (P:0.32001)
23. **Twenty-first Branch (Right from Node 22):**
* "[0] Therefore, the left uses 1(O) twice, for a total of 2(O). Second, we must find out how many oxygen molecules are needed to get a balanced equation. We balance by adding the same thing to both sides that can be multiplied the same on each side. In this" (Q:0.03162) (P:0.23481)
24. **Twenty-second Branch (Right from Node 23):**
* "[1] Next, we need to determine if CH4 is a limiting reagent. A limiting reagent is the reactant that is mostly likely to run out before all of the products can be made. This is important because we need to find out how many times we need to multiply the number of moles of oxygen" (Q:-0.01939) (P:0.38211)
### Key Observations
* The tree starts with identifying the imbalance in oxygen atoms.
* It explores different approaches to balancing, including adjusting coefficients and considering the number of atoms in each molecule.
* Several branches lead to similar conclusions about the number of oxygen molecules needed.
* The tree touches on the concept of limiting reagents, indicating a more advanced understanding of chemical reactions.
### Interpretation
The decision tree provides a step-by-step guide to balancing the combustion equation for methane. It highlights the importance of:
* Identifying the missing elements.
* Understanding the stoichiometry of the reaction.
* Adjusting coefficients to ensure the number of atoms is equal on both sides.
* Considering the role of limiting reagents.
The tree demonstrates a logical progression of thought, starting with basic concepts and gradually introducing more complex considerations. The presence of Q and P values at each node might represent some form of confidence or probability associated with each step, but without further context, their exact meaning is unclear. The tree effectively breaks down a complex task into manageable steps, making it easier to understand the process of balancing chemical equations.
DECODING INTELLIGENCE...