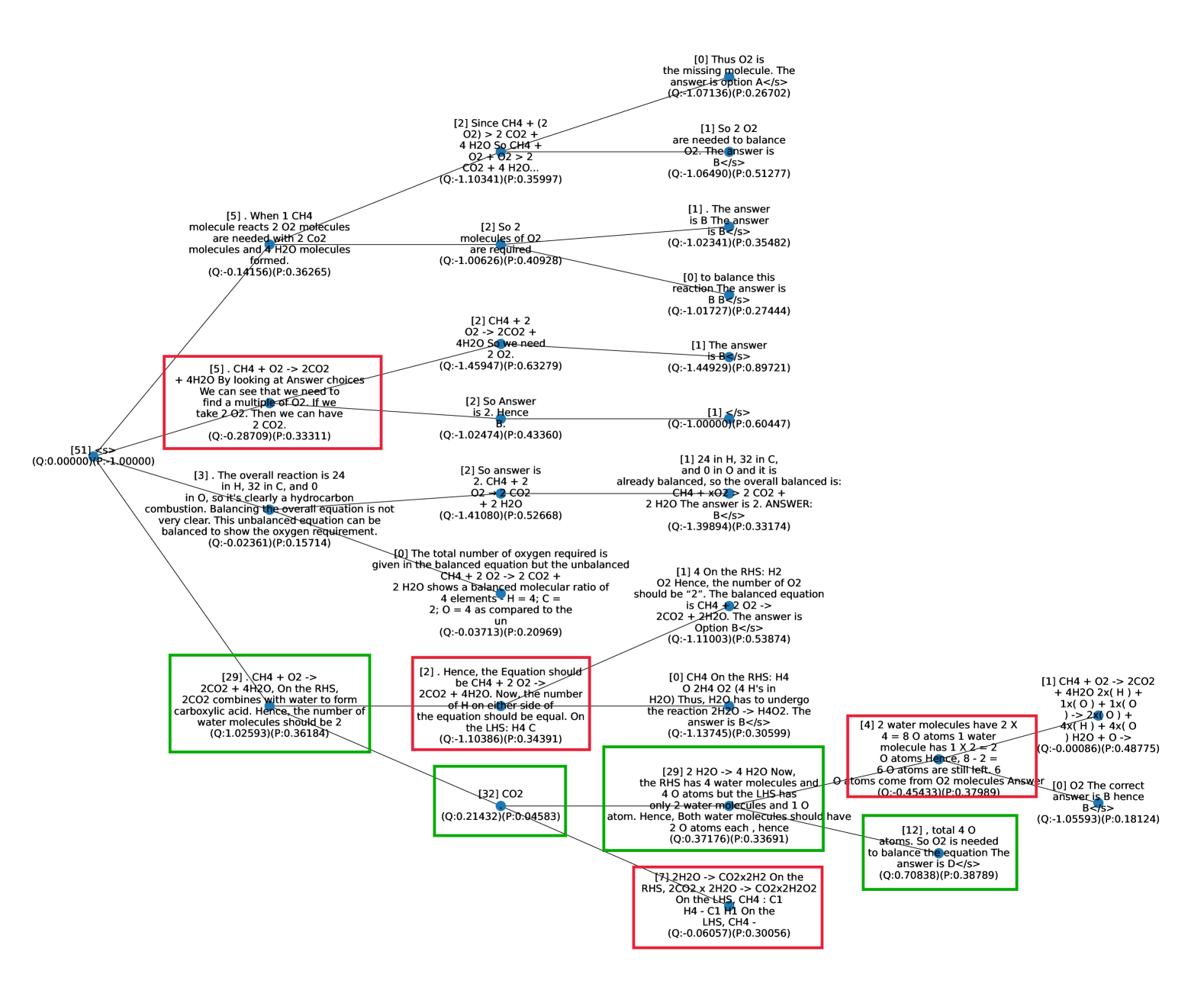
## Diagram: Chemical Reaction Network
### Overview
The image presents a network diagram illustrating different reasoning paths and statements related to balancing a chemical reaction, specifically the combustion of methane (CH4) with oxygen (O2) to produce carbon dioxide (CO2) and water (H2O). Each node contains a statement or deduction, along with associated Q and P values, likely representing some form of quality or probability scores. The diagram explores various approaches to understanding and balancing the chemical equation.
### Components/Axes
* **Nodes:** Each node contains a statement, question, or deduction related to the chemical reaction. Each node also contains a (Q:value)(P:value) pair.
* **Edges:** Arrows connect the nodes, indicating the flow of reasoning or logical connections between statements.
* **Textual Content:** The nodes contain various statements about the chemical reaction, including:
* Balancing the equation
* Identifying the missing molecule
* Determining the number of molecules required
* Analyzing the number of atoms on each side of the equation
* **Color Coding:**
* Red boxes surround statements related to the overall reaction and the need to find a multiple of O2.
* Green boxes surround statements related to the combination of CO2 with water to form carboxylic acid and the number of water molecules.
### Detailed Analysis or ### Content Details
Here's a breakdown of the content within selected nodes:
* **Node [51] (Top-Left):** (Q:0.00000)(P:-1.00000) - This node appears to be a starting point or a general reference.
* **Node [5] (Left):** "When 1 CH4 molecule reacts 2 O2 molecules are needed with 2 CO2 molecules and 4 H2O molecules formed." (Q:-0.14156)(P:0.36265)
* **Node [5] (Center-Left, Red Box):** "CH4 + O2 -> 2CO2 + 4H2O By looking at Answer choices We can see that we need to find a multiple of O2. If we take 2.O2. Then we can have 2 CO2." (Q:-0.28709)(P:0.33311)
* **Node [3] (Bottom-Left):** "The overall reaction is 24 in H, 32 in C, and 0 in O, so it's clearly a hydrocarbon combustion. Balancing the overall equation is not very clear. This unbalanced equation can be balanced to show the oxygen requirement." (Q:-0.02361)(P:0.15714)
* **Node [29] (Bottom-Left, Green Box):** "CH4 + O2 -> 2CO2 + 4H2O, On the RHS, 2CO2 combines with water to form carboxylic acid. Hence, the number of water molecules should be 2" (Q:1.02593)(P:0.36184)
* **Node [2] (Top):** "Since CH4 + (2 O2) > 2 CO2 + 4 H2O So CH4 + O2 + O2 > 2 CO2 + 4 H2O..." (Q:-1.10341)(P:0.35997)
* **Node [0] (Top-Right):** "Thus O2 is the missing molecule. The answer is option A</s>" (Q:-1.07136)(P:0.26702)
* **Node [1] (Top-Right):** "So 2 O2 are needed to balance O2. The answer is B</s>" (Q:-1.06490)(P:0.51277)
* **Node [1] (Center-Right):** "24 in H, 32 in C, and 0 in O and it is already balanced, so the overall balanced is: CH4 + xO2 -> 2 CO2 + 2 H2O The answer is 2. ANSWER: B</s>" (Q:-1.39894)(P:0.33174)
* **Node [32] (Bottom-Center):** "CO2" (Q:0.21432)(P:0:04583)
* **Node [2] (Center):** "So answer is 2. CH4 + 2 O2 -> 2 CO2 + 2 H2O" (Q:-1.41080)(P:0.52668)
### Key Observations
* The diagram explores different approaches to balancing the chemical equation for methane combustion.
* Several nodes focus on the number of oxygen molecules required for the reaction.
* The Q and P values associated with each node likely represent the quality or probability of the statement being correct.
* The color-coding highlights specific aspects of the reaction, such as the formation of carboxylic acid and the overall reaction requirements.
### Interpretation
The diagram represents a problem-solving process for balancing a chemical equation. It shows different lines of reasoning, some of which may be more accurate or efficient than others. The Q and P values could represent the confidence level or accuracy associated with each step in the reasoning process. The connections between nodes indicate how different ideas and deductions relate to each other in solving the problem. The red and green boxes highlight key aspects of the chemical reaction, such as the overall reaction requirements and the potential formation of carboxylic acid. The diagram suggests a multi-faceted approach to understanding and balancing chemical equations, where different perspectives and deductions contribute to the final solution.