TECHNICAL ASSET FINGERPRINT
5d9c534df5a72e8b476e0f06
Click to view fullscreen

Press ESC or click to close
FOUND IN PAPERS
EXPERT: gemini-2.0-flash VERSION 1
RUNTIME: nugit/gemini/gemini-2.0-flash
INTEL_VERIFIED
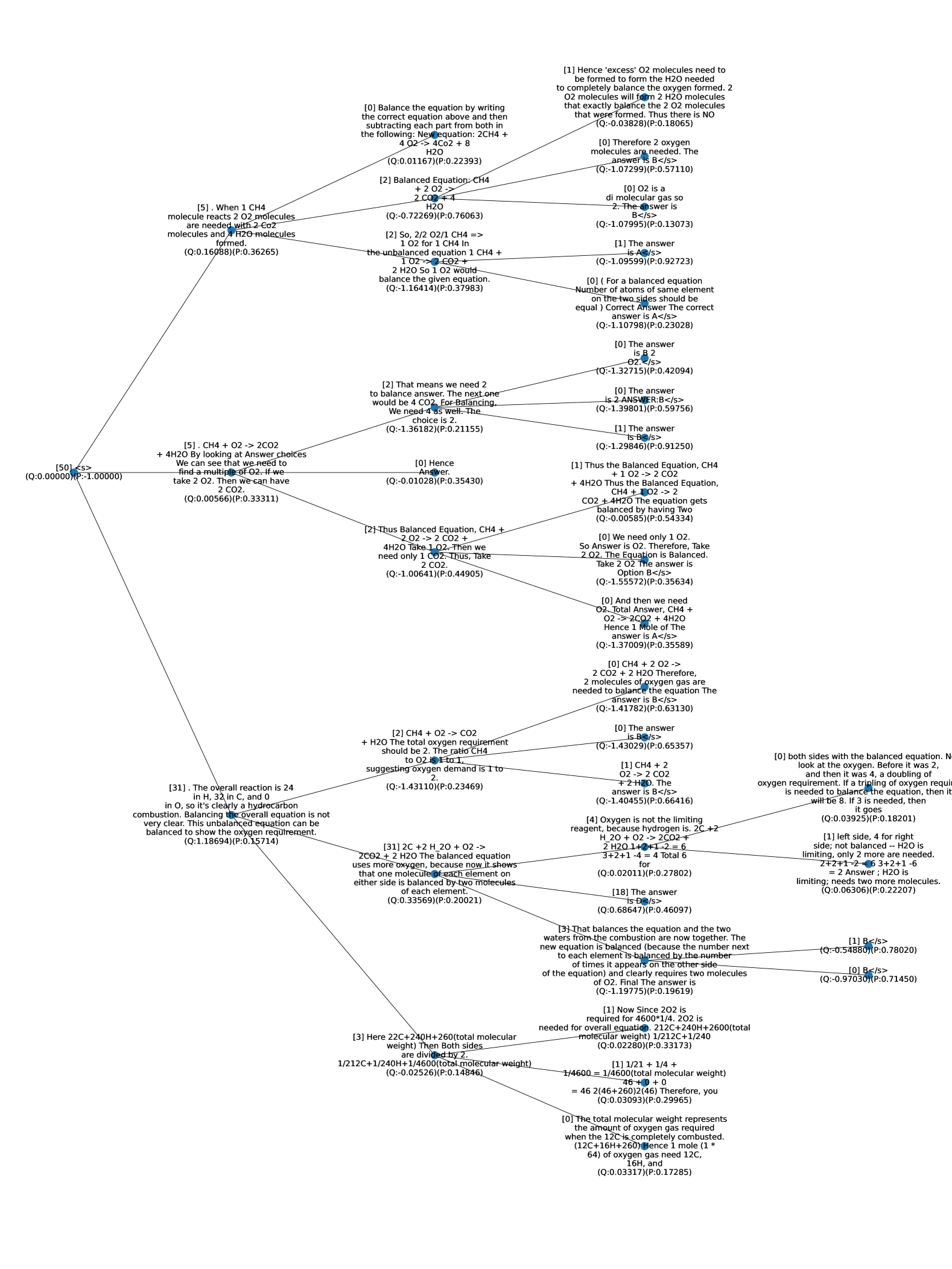
## Decision Tree: Chemical Equation Balancing
### Overview
The image depicts a decision tree diagram related to balancing a chemical equation, specifically the combustion of methane (CH4). The tree branches out based on different approaches and considerations for balancing the equation, ultimately leading to various conclusions and answers. Each node contains text explaining the reasoning and calculations involved, along with associated Q and P values (likely representing some form of score or probability).
### Components/Axes
* **Nodes:** Each node contains a statement or question related to the balancing process.
* **Branches:** Lines connecting the nodes, representing different paths of reasoning.
* **Q and P Values:** Each node has associated (Q:value)(P:value) pairs, which are numerical values. The meaning of Q and P is not explicitly defined in the image, but they likely represent a quality score and probability, respectively.
* **Root Node:** The starting point of the decision tree, labeled "[50] <s> (Q:0.00000)(P:-1.00000)".
* **Leaf Nodes:** The ending points of the branches, representing final answers or conclusions.
### Detailed Analysis or ### Content Details
Here's a breakdown of the tree's content, following the branching logic:
1. **Root Node:** "[50] <s> (Q:0.00000)(P:-1.00000)" - This is the starting point.
2. **First Branch:**
* Node: "[5]. When 1 CH4 molecule reacts 2 O2 molecules are needed with 2 CO2 molecules and 4 H2O molecules formed. (Q:0.16088)(P:0.36265)"
* Branch 1:
* Node: "[0] Balance the equation by writing the correct equation above and then subtracting each part from both in the following: New equation: 2CH4 + 4 O2 -> 4Co2 + 8 H2O (Q:0.01167)(P:0.22393)"
* Branch 1.1:
* Node: "[1] Hence 'excess' 02 molecules need to be formed to form the H2O needed to completely balance the oxygen formed. 2 02 molecules will form 2 H20 molecules that exactly balance the 2 02 molecules that were formed. Thus there is NO (Q:-0.03828)(P:0.18065)"
* Branch 1.1.1:
* Node: "[0] Therefore 2 oxygen molecules are needed. The answer is B</s> (Q:-1.07299)(P:0.57110)"
* Branch 1.1.2:
* Node: "[0] O2 is a di molecular gas so 2. The answer is B</s> (Q:-1.07995)(P:0.13073)"
* Branch 1.1.3:
* Node: "[1] The answer is A</s> (Q:-1.09599)(P:0.92723)"
* Branch 1.1.4:
* Node: "[0] (For a balanced equation Number of atoms of same element on the two sides should be equal) Correct Answer The correct answer is A</s> (Q:-1.10798)(P:0.23028)"
* Branch 1.1.5:
* Node: "[0] The answer is B 2 02.</s> (Q:-1.32715)(P:0.42094)"
* Branch 1.1.6:
* Node: "[0] The answer is 2 ANSWER:B</s> (Q:-1.39801)(P:0.59756)"
* Branch 1.1.7:
* Node: "[1] The answer is B</s> (Q:-1.29846)(P:0.91250)"
* Branch 2:
* Node: "[2] Balanced Equation: CH4 + 2 02-> 2 CO2 + 4 H2O (Q:-0.72269)(P:0.76063)"
* Branch 2.1:
* Node: "[1] Thus the Balanced Equation, CH4 + 1 02 -> 2 CO2 + 4H2O Thus the Balanced Equation, CH4 + 1 02 -> 2 CO2 + 4H2O The equation gets balanced by having Two (Q:-0.00585)(P:0.54334)"
* Branch 2.1.1:
* Node: "[1] B</s> (Q:-0.54880)(P:0.78020)"
* Branch 2.2:
* Node: "[2] So, 2/2 02/1 CH4 => 1 02 for 1 CH4 In the unbalanced equation 1 CH4 + 1 02-> 2 CO2 + 2 H2O So 1 02 would balance the given equation. (Q:-1.16414)(P:0.37983)"
* Branch 2.2.1:
* Node: "[0] We need only 1 02. So Answer is 02. Therefore, Take 2 02. The Equation is Balanced. Take 2 O2 The answer is Option B</s> (Q:-1.55572)(P:0.35634)"
* Branch 2.3:
* Node: "[2] That means we need 2 to balance answer. The next one would be 4 CO2 For Balancing. We need 4 as well. The choice is 2. (Q:-1.36182)(P:0.21155)"
* Branch 2.3.1:
* Node: "[0] And then we need 02. Total Answer, CH4 + 02-> 2CO2 + 4H2O Hence 1 Mole of The answer is A</s> (Q:-1.37009)(P:0.35589)"
* Branch 2.4:
* Node: "[0] Hence Answer. (Q:-0.01028)(P:0.35430)"
* Branch 2.4.1:
* Node: "[0] CH4 + 2 02 -> 2 CO2 + 2 H2O Therefore, 2 molecules of oxygen gas are needed to balance the equation The answer is B</s> (Q:-1.41782)(P:0.63130)"
* Branch 3:
* Node: "[5]. CH4 + O2 -> 2CO2 + 4H2O By looking at Answer choices We can see that we need to find a multiple of 02. If we take 2 02. Then we can have 2 CO2. (Q:0.00566)(P:0.33311)"
* Branch 3.1:
* Node: "[0] The answer is B</s> (Q:-1.43029)(P:0.65357)"
* Branch 4:
* Node: "[2] Thus Balanced Equation, CH4 + 2 02 -> 2 CO2 + 4H2O Take 1 02. Then we need only 1 CO2. Thus, Take 2 CO2. (Q:-1.00641)(P:0.44905)"
* Branch 4.1:
* Node: "[1] CH4 + 2 02 -> 2 CO2 + 2 H2O. The answer is B</s> (Q:-1.40455)(P:0.66416)"
* Branch 5:
* Node: "[2] CH4 + O2 -> CO2 + H2O The total oxygen requirement should be 2. The ratio CH4 to 02 is 1 to 1, suggesting oxygen demand is 1 to 2. (Q:-1.43110)(P:0.23469)"
* Branch 5.1:
* Node: "[4] Oxygen is not the limiting reagent, because hydrogen is. 2C +2 Η 20 + 02 -> 2CO2 + 2 H2O 1+2+1 -2 = 6 3+2+1-4 = 4 Total 6 for (Q:0.02011)(P:0.27802)"
* Branch 5.1.1:
* Node: "[1] left side, 4 for right side; not balanced -- H2O is limiting, only 2 more are needed. 2+2+1-2-6 3+2+1-6 = 2 Answer; H2O is limiting; needs two more molecules. (Q:0.06306)(P:0.22207)"
* Branch 6:
* Node: "[31] 2C +2 Η 20 + O2 -> 2CO2 + 2 H2O The balanced equation uses more oxygen, because now it shows that one molecule of each element on either side is balanced by two molecules of each element. (Q:0.33569)(P:0.20021)"
* Branch 6.1:
* Node: "[18] The answer is D</s> (Q:0.68647)(P:0.46097)"
* Branch 7:
* Node: "[31]. The overall reaction is 24 in H, 32 in C, and 0 in O, so it's clearly a hydrocarbon combustion. Balancing the overall equation is not very clear. This unbalanced equation can be balanced to show the oxygen requirement. (Q:1.18694)(P:0.15714)"
* Branch 7.1:
* Node: "[3] That balances the equation and the two waters from the combustion are now together. The new equation is balanced (because the number next to each element is balanced by the number of times it appears on the other side of the equation) and clearly requires two molecules of 02. Final The answer is (Q:-1.19775)(P:0.19619)"
* Branch 7.1.1:
* Node: "[1] B</s> (Q:-0.97030)(P:0.71450)"
* Branch 8:
* Node: "[3] Here 22C+240H+260(total molecular weight) Then Both sides are divided by 2. 1/212C+1/240H+1/4600(total molecular weight) (Q:-0.02526)(P:0.14846)"
* Branch 8.1:
* Node: "[1] Now Since 202 is required for 4600*1/4. 202 is needed for overall equation. 212C+240H+2600(total molecular weight) 1/212C+1/240 (Q:0.02280)(P:0.33173)"
* Branch 8.1.1:
* Node: "[1] B</s> (Q:-0.54880)(P:0.78020)"
* Branch 9:
* Node: "[1] 1/21 + 1/4 + 1/4600 = 1/4600(total molecular weight) 46 + 0 + 0 = 46 2(46+260)2(46) Therefore, you (Q:0.03093)(P:0.29965)"
* Branch 10:
* Node: "[0] The total molecular weight represents the amount of oxygen gas required when the 120 is completely combusted. (12C+16H+260) Hence 1 mole (1 * 64) of oxygen gas need 12C, 16H, and (Q:0.03317)(P:0.17285)"
* Branch 11:
* Node: "[0] both sides with the balanced equation. N look at the oxygen. Before it was 2, and then it was 4, a doubling of oxygen requirement. If a tripling of oxygen requis is needed to balance the equation, then it will be 8. If 3 is needed, then it goes (Q:0.03925)(P:0.18201)"
### Key Observations
* The tree explores various approaches to balancing the methane combustion equation.
* Many branches lead to the conclusion that "B" is the answer, although the meaning of "B" is not explicitly defined. It likely refers to a specific answer choice in a multiple-choice question.
* The Q and P values vary across the nodes, potentially indicating the confidence or accuracy associated with each step in the reasoning.
* Some branches involve more complex calculations and considerations than others.
### Interpretation
The decision tree illustrates the different paths one might take when balancing a chemical equation. It highlights the importance of understanding stoichiometry and the relationships between reactants and products. The tree also suggests that there may be multiple ways to arrive at the correct answer, depending on the approach taken. The Q and P values could be used to assess the reliability of each path, with higher values indicating more confident or accurate reasoning. The frequent appearance of "B" as the answer suggests that this is the correct choice based on the given context and answer options.
DECODING INTELLIGENCE...