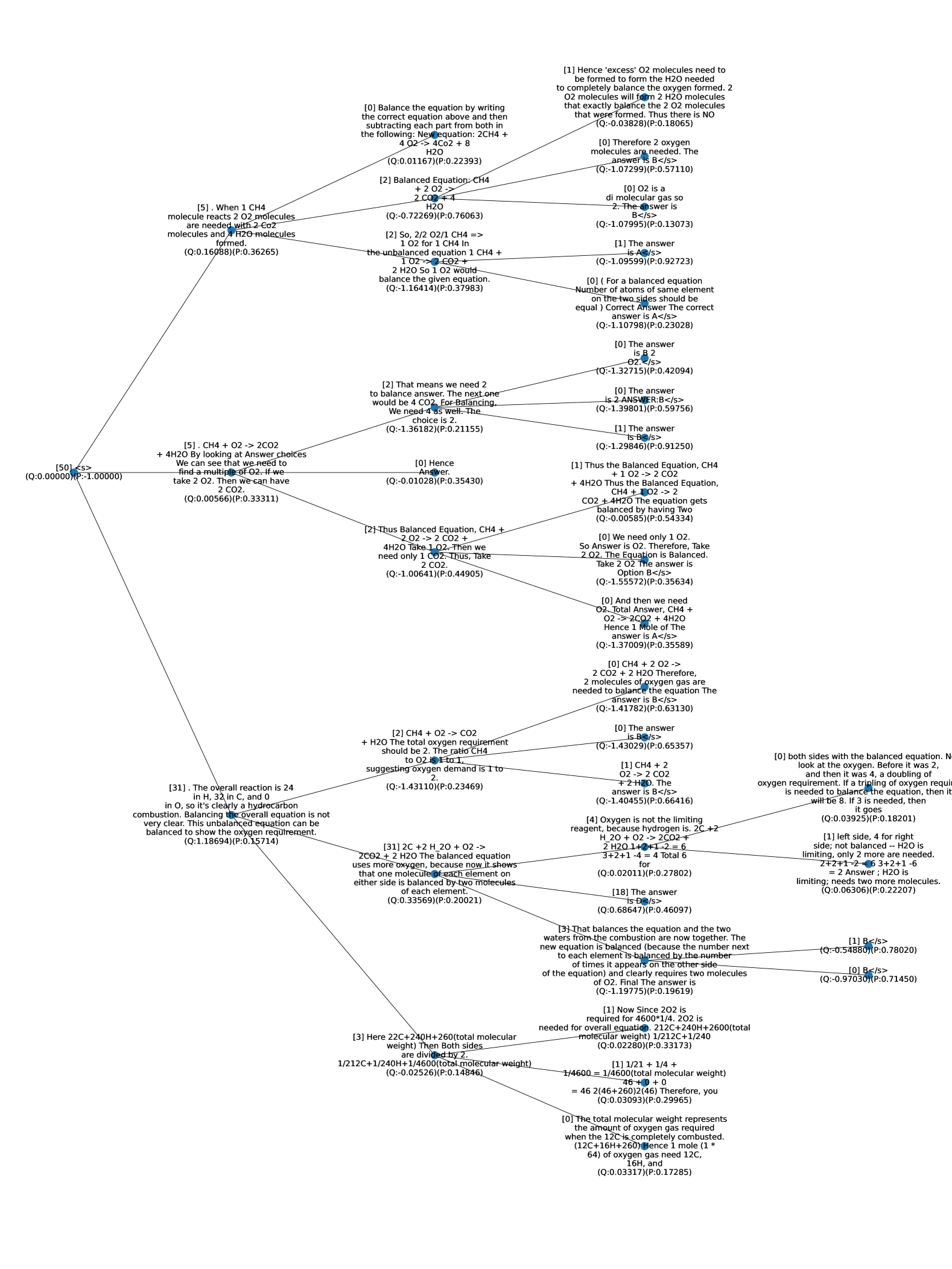
## Diagram: Balancing Chemical Equations - Flowchart
### Overview
This image presents a flowchart detailing the steps involved in balancing chemical equations. The flowchart guides the user through a series of questions and calculations to arrive at the correct stoichiometric coefficients. The diagram is complex, with numerous decision points and calculations presented in labeled boxes connected by arrows. The flowchart is oriented vertically, with the starting point at the top and the ending point at the bottom.
### Components/Axes
The diagram consists of interconnected boxes containing text and equations. The boxes are numbered sequentially, indicating the order of steps. Arrows indicate the flow of logic. There are no traditional axes or scales. The flowchart is divided into sections based on the type of equation being balanced (e.g., combustion, general).
### Detailed Analysis or Content Details
Here's a breakdown of the flowchart's content, following the flow from top to bottom:
**Starting Point (Top):**
* "[S] Is CH4 a reactant?" (0.000000[P:0.000000]) - Leads to "Yes" branch.
* "[S] Is O2 a reactant?" (0.000000[P:0.000000]) - Leads to "Yes" branch.
**Combustion Branch (Top-Left):**
* "[1] When 1 CH4 molecule is burned, how many O2 molecules are needed?" (0.16[P:0.36265])
* "[2] That means we need 2 O2 molecules for 1 CH4 molecule. We now have: CH4 + 2O2 ->" (0.000000[P:0.000000])
* "[3] Balance Carbon: CH4 + 2O2 -> CO2 + H2O" (0.000000[P:0.000000])
* "[4] Balance Hydrogen: CH4 + 2O2 -> CO2 + 2H2O" (0.000000[P:0.000000])
* "[5] Balance Oxygen: CH4 + 2O2 -> CO2 + 2H2O" (0.000000[P:0.000000])
* "[6] The answer: CH4 + 2O2 -> CO2 + 2H2O" (0.000000[P:0.000000])
**General Equation Branch (Top-Right):**
* "[1] Hence "excess" O2 molecules need to completely balance the oxygen formed. 2 H2 + O2 -> 2 H2O. Thus, the NO. of O2 molecules is 1." (0.10729[P:0.57110])
* "[0] Therefore, 2 oxygen molecules are needed." (0.10729[P:0.57110])
* "[0] Is O2 a reactant?" (0.107995[P:1.3073])
* "[1] The answer" (0.107995[P:1.3073])
* "[0] [1] In a balanced equation the number of atoms of each element on both sides of the equation must be equal. " (0.107988[P:1.2038])
* "[0] The answer" (0.107988[P:1.2038])
* "[0] [3] The answer" (0.107988[P:0.42094])
* "[1] The answer" (0.1380[P:0.59756])
* "[0] [5] The answer" (0.1380[P:0.59756])
* "[0] Balance the equation by writing the correct coefficients in front of each molecule. 2 C2H6 + 7 O2 -> 4 CO2 + 6 H2O" (0.1380[P:0.59756])
* "[0] Balance the equation: 2 C2H6 + 7 O2 -> 4 CO2 + 6 H2O" (0.1380[P:0.59756])
* "[0] That means Carbon = 4, Hydrogen = 12, Oxygen = 14" (0.1380[P:0.59756])
* "[0] [7] The answer" (0.1380[P:0.59756])
* "[0] Balance the equation: N2 + 3 H2 -> 2 NH3" (0.1380[P:0.59756])
* "[0] That means Nitrogen = 2, Hydrogen = 6" (0.1380[P:0.59756])
* "[0] [9] The answer" (0.1380[P:0.59756])
**Central Branch (Middle):**
* "[S] Is it a combustion reaction?" (0.000000[P:0.000000]) - Leads to "Yes" and "No" branches.
* "[0] Balance the equation by writing the correct coefficients in front of each molecule. 4 NH3 + 5 O2 -> 4 NO + 6 H2O" (0.333333[P:0.4535])
* "[0] Balance the equation: 4 NH3 + 5 O2 -> 4 NO + 6 H2O" (0.333333[P:0.4535])
* "[0] That means Nitrogen = 4, Hydrogen = 12, Oxygen = 10" (0.333333[P:0.4535])
* "[0] [11] The answer" (0.333333[P:0.4535])
**Bottom Section:**
* "Balancing Chemical Equations" (Title)
* "Dr. B. N. Jagtap, Dept. of Chemistry, A. C. Patil College of Arts, Science & Commerce, Osamanabad" (Author/Affiliation)
* "www.jagtapscience.com" (Website)
The values in brackets "[P:value]" appear consistently throughout the diagram. These likely represent probabilities or confidence levels associated with each step or answer.
### Key Observations
* The flowchart is highly structured and methodical, breaking down the balancing process into discrete steps.
* The use of questions ("Is CH4 a reactant?") guides the user through the appropriate path.
* The inclusion of example equations (e.g., CH4 + 2O2 -> CO2 + 2H2O) helps illustrate the process.
* The "[P:value]" notation suggests a probabilistic or confidence-based approach to the balancing process, though the meaning of these values is not explicitly defined.
* The diagram focuses on combustion reactions and general balancing techniques.
### Interpretation
This flowchart serves as a pedagogical tool for teaching students how to balance chemical equations. It emphasizes a step-by-step approach, starting with identifying reactants and then systematically balancing each element. The flowchart's structure promotes logical thinking and problem-solving skills. The inclusion of example equations and the "[P:value]" notation suggest a nuanced understanding of the balancing process, acknowledging that there may be multiple valid solutions or varying degrees of confidence in the final answer. The author's affiliation and website indicate a commitment to providing accessible educational resources in chemistry. The diagram is a clear and concise representation of a complex scientific concept, making it a valuable learning aid. The consistent use of "[P:value]" suggests a potentially more advanced or statistical approach to balancing, which is not fully explained within the diagram itself.