\n
## Diagram: Cyclohexane Conformers
### Overview
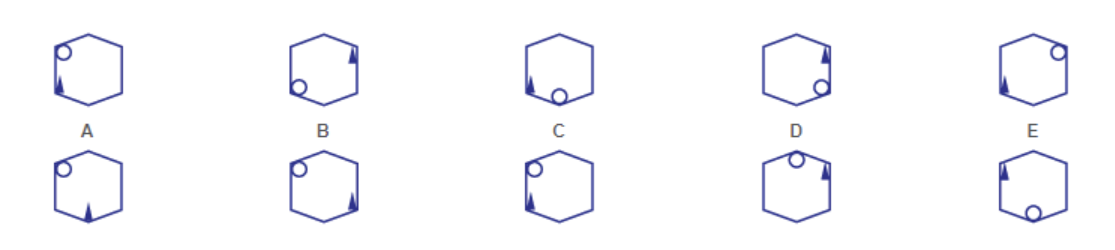
The image displays a series of cyclohexane ring diagrams, representing different conformational isomers. Each diagram depicts a six-membered carbon ring with a single substituent (represented by a small circle) attached to one of the carbon atoms. The diagrams are labeled A through E, with two rows of diagrams each.
### Components/Axes
The image consists of cyclohexane ring diagrams. There are no axes or legends in the traditional sense. The labels A, B, C, D, and E identify each individual diagram. Each diagram shows a cyclohexane ring with a single substituent.
### Detailed Analysis or Content Details
The diagrams represent different chair conformations of cyclohexane. The substituent's position varies in each diagram.
* **A:** The substituent is in an equatorial position in the top row and axial in the bottom row.
* **B:** The substituent is in an axial position in the top row and equatorial in the bottom row.
* **C:** The substituent is in an equatorial position in both the top and bottom rows.
* **D:** The substituent is in an axial position in both the top and bottom rows.
* **E:** The substituent is in an equatorial position in the top row and axial in the bottom row.
The cyclohexane rings are drawn in a "zig-zag" style, representing the chair conformation. The substituent is represented by a small circle.
### Key Observations
The diagrams illustrate the concept of chair flipping in cyclohexane. The substituent can switch between axial and equatorial positions. Diagrams A, B, and E show the chair flip, while C and D show the substituent remaining in the same position (equatorial or axial, respectively).
### Interpretation
The image demonstrates the dynamic nature of cyclohexane conformations. The chair conformation is not static; it can interconvert to another chair conformation through a process called chair flipping. This process involves the axial and equatorial substituents switching positions. Equatorial positions are generally more stable than axial positions due to reduced steric interactions. The image highlights the different possible conformations and the ability of the substituent to occupy either an axial or equatorial position. The image is a visual aid for understanding conformational analysis in organic chemistry.