## Technical Document: Inorganic Synthesis Experiment Workflow and Data Analysis
### Overview
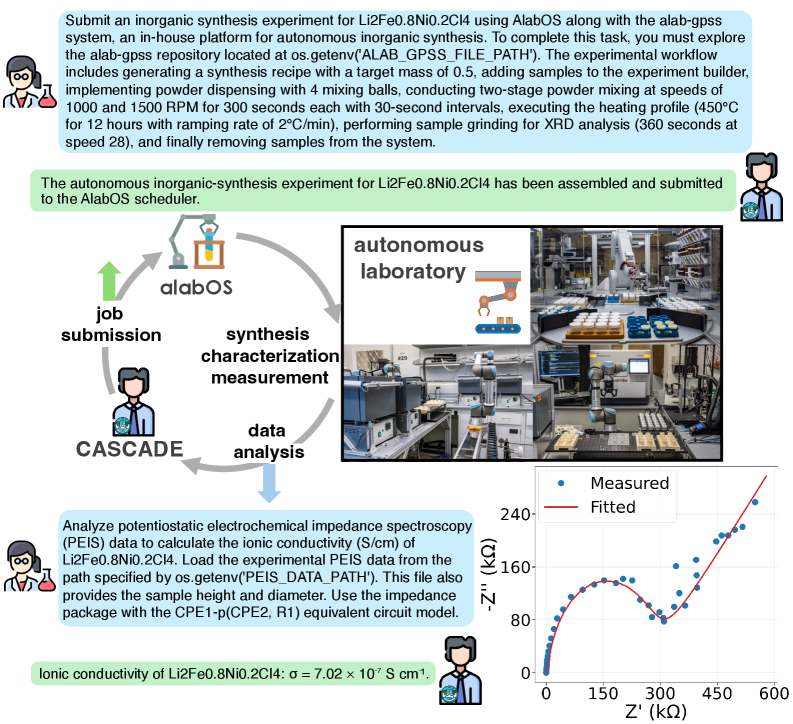
The image depicts a technical workflow for an autonomous inorganic synthesis experiment using the AlabOS platform and alab-gpss repository. It includes a flowchart of the experimental process, a graph of electrochemical impedance spectroscopy (EIS) data, and descriptions of laboratory automation. Key elements include synthesis parameters, data analysis steps, and a plot of ionic conductivity.
---
### Components/Axes
#### Text Blocks
1. **Experiment Submission Instructions**:
- Target: Li2Fe0.8Ni0.2Cl4 synthesis
- Workflow:
- Generate synthesis recipe with 0.5g target mass
- Implement powder dispensing (4 mixing balls, 1000/1500 RPM, 300s each)
- Two-stage mixing at 450°C (30s intervals, 12h total)
- Sample grinding for XRD (360s at 28°C/min)
- Sample removal
- Submission status: "The autonomous inorganic-synthesis experiment for Li2Fe0.8Ni0.2Cl4 has been assembled and submitted to the AlabOS scheduler."
2. **Data Analysis Instructions**:
- Analyze PEIS data to calculate ionic conductivity (S/cm)
- Load experimental PEIS data from `os.getenv('PEIS_DATA_PATH')`
- Use impedance package with CPE1-p(CPE2, R1) equivalent circuit model
- Result: Ionic conductivity of Li2Fe0.8Ni0.2Cl4: σ = 7.02×10⁻⁷ S/cm
#### Diagram (Flowchart)
- **Components**:
- Job submission (human icon)
- AlabOS platform (central node)
- Synthesis (flask icon)
- Characterization (microscope icon)
- Measurement (spectrometer icon)
- Data analysis (computer icon)
- Cascade system (loop arrow)
- **Flow**:
- Job submission → AlabOS → Synthesis → Characterization → Measurement → Data analysis → Cascade
#### Graph (EIS Data)
- **Axes**:
- X-axis: Z' (kΩ) [0–600]
- Y-axis: -Z'' (kΩ) [0–240]
- **Legend**:
- Blue dots: Measured data
- Red line: Fitted curve
- **Key Data Point**:
- Ionic conductivity: σ = 7.02×10⁻⁷ S/cm (annotated in text)
#### Laboratory Image
- **Components**:
- Robotic arms (blue/gray)
- Sample holders (yellow/orange)
- Automated equipment (conveyors, grinders)
- Control panels (monitors, buttons)
---
### Detailed Analysis
#### Text Blocks
- **Synthesis Parameters**:
- Target mass: 0.5g
- Mixing: 4 balls, 1000/1500 RPM, 300s per stage
- Temperature: 450°C (30s intervals, 12h total)
- Grinding: 360s at 28°C/min ramping
- **Data Analysis**:
- PEIS data path: `os.getenv('PEIS_DATA_PATH')`
- Sample dimensions: Height/diameter provided
- Circuit model: CPE1-p(CPE2, R1)
#### Graph
- **Trend**:
- Measured data (blue dots) shows a U-shaped curve with a minimum at ~200 kΩ
- Fitted curve (red line) closely follows the data, with slight deviations
- **Key Observation**:
- Ionic conductivity (σ) derived from EIS data: 7.02×10⁻⁷ S/cm
---
### Key Observations
1. The synthesis workflow emphasizes precision (e.g., 30s intervals, 2°C/min ramping).
2. The EIS graph shows a typical semicircular arc, indicating charge transfer resistance and diffusion processes.
3. The ionic conductivity value (7.02×10⁻⁷ S/cm) suggests moderate ionic mobility in the synthesized material.
---
### Interpretation
- **Workflow Integration**: The AlabOS platform automates synthesis and data collection, reducing human intervention. The Cascade system ensures iterative refinement.
- **Material Properties**: The ionic conductivity value (7.02×10⁻⁷ S/cm) indicates the material’s potential for applications requiring ion transport (e.g., batteries, sensors).
- **Data Validation**: The close match between measured and fitted EIS data validates the equivalent circuit model (CPE1-p(CPE2, R1)).
- **Automation Impact**: Robotic systems (e.g., powder dispensing, sample grinding) enable reproducibility and scalability of experiments.
---
### Notable Trends
- The EIS curve’s minimum at ~200 kΩ suggests optimal ionic conductivity at this frequency.
- The synthesis parameters (high temperature, prolonged mixing) likely enhance material homogeneity, reflected in the conductivity data.