TECHNICAL ASSET FINGERPRINT
ae87af93e5d63973c9e7d7de
Click to view fullscreen

Press ESC or click to close
FOUND IN PAPERS
EXPERT: healer-alpha-free VERSION 1
RUNTIME: free/openrouter/healer-alpha
INTEL_VERIFIED
\n
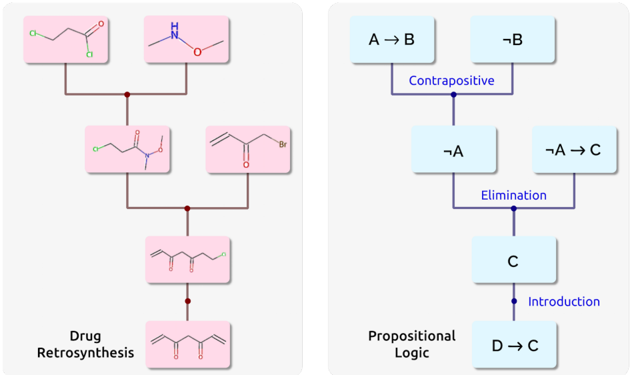
## Dual Diagram: Drug Retrosynthesis and Propositional Logic Flowchart
### Overview
The image displays two distinct, side-by-side diagrams on a light gray background. The left panel illustrates a chemical "Drug Retrosynthesis" pathway using molecular structures in pink boxes. The right panel depicts a "Propositional Logic" deduction flowchart using text in light blue boxes. Both diagrams show a top-to-bottom flow of steps, connected by lines and labeled operations.
### Components/Axes
**Left Panel (Drug Retrosynthesis):**
* **Title:** "Drug Retrosynthesis" (bottom-left).
* **Components:** Six pink rectangular boxes containing 2D chemical structure diagrams. They are connected by a brown branching line structure indicating synthetic steps.
* **Flow:** The pathway flows from two starting materials at the top, converging and reacting with a third compound, leading to a final target molecule at the bottom.
**Right Panel (Propositional Logic):**
* **Title:** "Propositional Logic" (bottom-center).
* **Components:** Six light blue rectangular boxes containing logical propositions and operations. They are connected by dark blue lines.
* **Flow:** The deduction flows from two initial premises at the top, through three labeled logical operations ("Contrapositive", "Elimination", "Introduction"), to a final conclusion at the bottom.
### Detailed Analysis
#### **1. Drug Retrosynthesis (Left Panel)**
* **Step 1 (Top Row):**
* **Left Box:** A molecule with a chloro group (Cl) attached to a three-carbon chain ending in a methyl ester (COOCH₃). Structure: Cl-CH₂-CH₂-COOCH₃.
* **Right Box:** A molecule with a secondary amine (NH) attached to a methoxy group (OCH₃). Structure: CH₃-NH-OCH₃ (methoxy(methyl)amine).
* **Connection:** A line descends from each box, converging at a central node.
* **Step 2 (Middle-Top Box):**
* The product of Step 1. A molecule where the amine nitrogen from the right box is now bonded to the carbonyl carbon of the ester from the left box, forming an N-methoxy-N-methyl amide (Weinreb amide). Structure: Cl-CH₂-CH₂-CO-N(CH₃)-OCH₃.
* **Connection:** A line descends from this box to a central node.
* **Step 3 (Middle-Right Box):**
* A separate reactant introduced at this stage. An α,β-unsaturated ketone with a bromomethyl group. Structure: CH₂=CH-CO-CH₂-Br (4-bromo-3-buten-2-one).
* **Connection:** A line descends from this box to the same central node as Step 2.
* **Step 4 (Middle-Bottom Box):**
* The product of Steps 2 & 3. A larger molecule combining the two precursors. It appears to be a 1,5-dicarbonyl compound with a terminal alkene and a bromine atom. Structure: CH₂=CH-CO-CH₂-CH₂-CO-N(CH₃)-OCH₃.
* **Connection:** A single line descends to the final box.
* **Step 5 (Bottom Box - Target Molecule):**
* The final target of the retrosynthesis. A symmetrical molecule with two terminal alkene groups and two ketone groups in the center. Structure: CH₂=CH-CO-CH₂-CH₂-CO-CH=CH₂ (1,6-diphenyl-1,5-hexadiene-3,4-dione is a possible analog, but the drawn structure is aliphatic: 1,5-hexadiene-3,4-dione).
#### **2. Propositional Logic (Right Panel)**
* **Initial Premises (Top Row):**
* **Left Box:** "A → B" (If A, then B).
* **Right Box:** "¬B" (Not B).
* **First Operation:**
* A line connects the two top boxes to a node labeled **"Contrapositive"**.
* **Result (Box below node):** "¬A" (Not A). This is the valid contrapositive inference from "A → B" and "¬B".
* **Second Operation:**
* A new premise is introduced in a box to the right: "¬A → C" (If not A, then C).
* A line connects the "¬A" box and the "¬A → C" box to a node labeled **"Elimination"** (also known as Modus Ponens).
* **Result (Box below node):** "C". This is the valid inference from "¬A" and "¬A → C".
* **Third Operation:**
* A line descends from the "C" box to a node labeled **"Introduction"** (likely referring to the rule of Conditional Introduction or a similar proof step).
* **Final Conclusion (Bottom Box):** "D → C" (If D, then C). This suggests that within the broader logical argument not fully shown, the truth of C has been established, allowing the derivation of a conditional statement involving a new proposition D.
### Key Observations
1. **Parallel Structure:** Both diagrams use a similar visual metaphor—a top-down flowchart with labeled operations—to represent complex, stepwise processes in two very different domains (chemistry and logic).
2. **Chemical Specificity:** The retrosynthesis pathway is concrete, showing specific molecular structures and functional group transformations (ester to Weinreb amide, subsequent carbon-carbon bond formation).
3. **Logical Abstraction:** The logic diagram is entirely abstract, using propositional variables (A, B, C, D) and standard inference rules. The final step ("D → C") implies a context beyond the visible premises.
4. **Visual Design:** The chemistry diagram uses organic, structural drawings in warm pink, while the logic diagram uses clean, textual statements in cool blue, creating a clear visual distinction.
### Interpretation
This image juxtaposes two fundamental types of systematic reasoning:
* **Drug Retrosynthesis** represents **practical, synthetic problem-solving**. It works backward from a desired complex molecule (the target) to identify simpler, commercially available or synthetically accessible starting materials. The diagram shows a plausible route involving amide formation and a conjugate addition/alkylation step. The presence of the Weinreb amide is notable, as it's a common strategy for controlled ketone synthesis.
* **Propositional Logic** represents **formal, deductive reasoning**. It demonstrates how new truths (conclusions) are rigorously derived from accepted premises using validated rules of inference. The flow is airtight: from ¬B and A→B, ¬A is deduced; from ¬A and ¬A→C, C is deduced. The final "Introduction" step is less self-contained, hinting at a larger proof where establishing C allows one to assert a conditional statement "D → C".
**Together, the diagrams highlight that both cutting-edge drug discovery and rigorous philosophical/mathematical thought rely on structured, stepwise deconstruction and reconstruction.** One manipulates physical matter, the other manipulates truth values, but both follow logical pathways to reach a goal. The chemistry side is a specific instance of a general strategy, while the logic side is a pure demonstration of form. The image serves as a visual metaphor for the underlying logical architecture present in both scientific and abstract disciplines.
DECODING INTELLIGENCE...