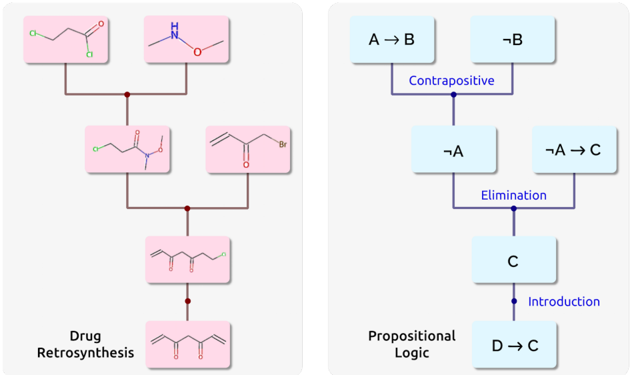
## Diagram: Drug Retrosynthesis and Propositional Logic Flowcharts
### Overview
The image contains two distinct flowcharts. The left diagram illustrates **Drug Retrosynthesis**, showing chemical structure transformations. The right diagram represents **Propositional Logic**, depicting logical deductions and rules. Both use labeled boxes connected by directional arrows.
---
### Components/Axes
#### Left Diagram (Drug Retrosynthesis):
- **Structure Boxes**:
1. Top-left: `O-C(=O)-C(=O)-O` (green and red atoms).
2. Top-right: `H-N-O` (blue nitrogen, red oxygen).
3. Middle-left: `O-C(=O)-C(=O)-N` (green, red, blue atoms).
4. Middle-right: `Br-C(=O)-C(=O)` (brown bromine, red oxygen).
5. Bottom: `O-C(=O)-C(=O)-C(=O)-O` (red oxygen, carbon chain).
- **Flow**:
- Top two boxes split into middle boxes via brown arrows.
- Middle boxes merge into the bottom box.
- **Legend**: None explicitly labeled.
#### Right Diagram (Propositional Logic):
- **Logical Statements**:
1. `A → B` (top-left).
2. `¬B` (top-right).
3. `¬A` (middle-left).
4. `¬A → C` (middle-right).
5. `C` (bottom-center).
6. `D → C` (bottom-left).
- **Arrows/Labels**:
- `A → B` → `¬A` via "Contrapositive" (purple arrow).
- `¬A → C` → `C` via "Elimination" (purple arrow).
- `D → C` → `C` via "Introduction" (blue arrow).
- **Legend**:
- Blue boxes for statements, purple arrows for rules.
- Labels in black text.
---
### Detailed Analysis
#### Left Diagram:
- **Chemical Transformations**:
- Initial structures decompose into intermediates (e.g., `O-C(=O)-C(=O)-N` and `Br-C(=O)-C(=O)`).
- Final product: A dicarboxylic acid derivative (`O-C(=O)-C(=O)-C(=O)-O`).
- **Uncertainties**:
- No explicit reaction conditions (e.g., temperature, catalysts) or yields provided.
#### Right Diagram:
- **Logical Deductions**:
1. From `A → B` and `¬B`, derive `¬A` (contrapositive).
2. From `¬A → C`, derive `C` (elimination).
3. From `D → C`, introduce `C` (introduction).
- **Rules**:
- Contrapositive: `A → B` ≡ `¬B → ¬A`.
- Elimination: `¬A → C` and `¬A` → `C`.
- Introduction: `D → C` and `D` → `C`.
---
### Key Observations
1. **Drug Retrosynthesis**:
- The process involves stepwise decomposition of a complex molecule into simpler precursors.
- Bromine (`Br`) and nitrogen (`N`) atoms are critical intermediates.
2. **Propositional Logic**:
- Logical rules (contrapositive, elimination, introduction) govern the flow of deductions.
- `C` is the terminal conclusion derived from multiple premises.
---
### Interpretation
- **Drug Retrosynthesis**:
- The flowchart mirrors organic synthesis planning, where a target molecule is broken down into accessible precursors. The presence of bromine and nitrogen suggests functional group manipulations (e.g., halogenation, amide formation).
- **Propositional Logic**:
- The diagram formalizes deductive reasoning. For example, `A → B` and `¬B` logically necessitate `¬A`, demonstrating modus tollens. The introduction of `C` via `D → C` highlights how new premises can validate conclusions.
- **Cross-Diagram Insight**:
- Both diagrams emphasize stepwise transformations: chemical structures in retrosynthesis and logical propositions in deduction. Each step depends on prior components, reflecting hierarchical dependencies.
- **Missing Data**:
- No quantitative metrics (e.g., reaction efficiency, logical validity scores) are provided, limiting empirical analysis.
---
**Note**: The diagrams are conceptual and lack numerical data. They serve as schematic representations of processes rather than empirical datasets.