# Natural statistics of binaural sounds
## Natural statistics of binaural sounds
Wiktor M lynarski ∗ 1 and J¨ urgen Jost 1,2
1 Max-Planck Institute for Mathematics in the Sciences, Leipzig, Germany 2 Santa Fe Institute, Santa Fe, New Mexico, USA
April 19, 2022
## Abstract
Binaural sound localization is usually considered a discrimination task, where interaural time (ITD) and level (ILD) disparities at pure frequency channels are utilized to identify a position of a sound source. In natural conditions binaural circuits are exposed to a stimulation by sound waves originating from multiple, often moving and overlapping sources. Therefore statistics of binaural cues depend on acoustic properties and the spatial configuration of the environment. In order to process binaural sounds efficiently, the auditory system should be adapted to naturally encountered cue distributions. Statistics of cues encountered naturally and their dependence on the physical properties of an auditory scene have not been studied before. Here, we performed binaural recordings of three auditory scenes with varying spatial properties. We have analyzed empirical cue distributions from each scene by fitting them with parametric probability density functions which allowed for an easy comparison of different scenes. Higher order statistics of binaural waveforms were analyzed by performing Independent Component Analysis (ICA) and studying properties of learned basis functions. Obtained results can be related to known neuronal mechanisms and suggest how binaural hearing can be understood in terms of adaptation to the natural signal statistics.
## Introduction
The idea that sensory systems reflect the statistical structure of stimuli encountered by organisms in their ecological niches [4, 3, 44] has driven numerous theoretical and experimental studies. Obtained results suggest that tuning properties of sensory neurons match regularities present in natural stimuli [46]. In light of this theory, neural representations, coding mechanisms and anatomical structures could be undestood by studying characteristics of the sensory environment.
To date, natural scene statistics research have been focusing mostly on visual stimuli [27]. Nevertheless, a number of interesting results relating natural sound statistics to the auditory system have also been delivered. For instance, Rieke et al demonstrated that auditory neurons in the frog increase information transmission, when the spectrum of the white-noise stimulus is shaped to match the spectrum of a frog call [43]. In a more recent experiment, Hsu and colleagues [26] have shown similar facilitation effects in the zebra finch auditory system using stimuli with power and phase modulation spectrum of a conspecific song. In a statistical study it has been shown that modulation spectra of natural sounds display a characteristic statistical
∗ Corresponding author. Email: mlynar@mis.mpg.de
signature [47] which allowed to form quantitative predictions about neural representations and coding of sounds. Other statistical models of natural auditory scenes have also led to interesting observations. Low-order, marginal statistics of amplitude envelopes, for instance, seem to be preserved across frequency channels as shown by Attias and Schreiner [2]. This means that all locations along the cochlea may be exposed to (on average) similar stimulation patterns in the natural environment. A strong evidence of adaptation of the early auditory system to natural sounds was provided by two complementary studies by Lewicki [32] and Smith and Lewicki [48]. The authors modeled high order statistics of natural stimuli by learning sparse representations of short sound chunks. In such a way, they reproduced filter shapes of the cat's cochlear nerve. These results were recently extended by Carlson et al [13] who obtained features resembling spectro-temporal receptive fields in the cat's Inferior Colliculus by learning sparse codes of speech spectrograms. Human perceptual capabilities have also been related to natural sound statistics in a recent study by McDermott and Simoncelli [37]. In a series of psychophysical experiments the authors have shown that perceived realism and recognizability of sound 'textures' by human subjects depends on how well the time-averaged statistics of stimulus modulation correspond to those of natural sounds. The aquired body of evidence strongly suggests that neural representations of acoustic stimuli reflect structures present in the natural auditory environment.
The above mentioned studies investigated statistical properties of single channel, monaural sounds relating them to the functioning of the nervous system. However, in natural hearing conditions the sensory input is determined by many additional factors - not only properties of the sound source. Air pressure waveforms reaching the cochlea are affected by positions and motion patterns of sound sources as well as head movements of the listening subject. These spatial aspects generate differences between stimuli present in each ear , which are traditionally divided into two classes: interaural level and phase differences [21]. The sound wavefront reaches firstly the ipsilateral ear and after a very short time delay the contralateral one. This generates the interaural time difference (ITD). After cochlear filtering - in pure frequency channels, ITDs correspond to phase differences (IPDs). Additionally, sound received by the contralateral ear is attenuated by the head, which generates the interaural level difference (ILD). According to the widely acknowdledged duplex theory [42, 21], in mammals, IPDs are used to localize low frequency sounds. The theory predicts that in higher frequency regimes IPDs become ambiguous and therefore sounds of frequency above a certain threshold (around 1 . 5 kHz in humans) are localized based on ILDs which become more pronounced due to the low-pass filtering properties of the head. Binaural cues are of a relative nature and positions of auditory objects are not represented on the sensory epiphelium - the cochlear membrane - in a direct way. They are reflected in binaural cue values, which themselves vary with changing spatial configuration of the environment and depend on sound sources' spectra.
Binaural hearing mechanisms have also been studied in terms of adaptation to natural stimulus statistics. Harper and McAlpine [23] have shown that tuning properties of IPD sensitive neurons in a number of species can be predicted from distributions of this cue naturally encountered by the organism. This was done by forming a model neuronal representation of maximal sensitivity to the stimulus change, as quantified by the Fisher information. Two recent experimental studies revealed rapid adaptation of binaural neurons and perceptual mechanisms to changing cue statistics. Dahmen and colleagues [14] stimulated human and animal subjects with non-stationary ILD sequences. They collected electrophysiological and psychophysical evidence in favor of adaptation to the stimulus distribution. Maier et al [33], in turn, have shown that neural tuning curves in the guinea pig and human performance in a localization task can be adapted to varying ITD distributions. Both - neural representation and human performance were, however, constrained to represent midline locations with the highest accuracy. One has to
note that Maier et al, take an issue with the interpretation of results obtained by Dahmen et al. suggesting that they may be explained by adaptation to the sound level and not ILDs per se.
Adaptation of the binaural auditory system to changes in the cue distribution occuring on different timescales seems to be experimentally confirmed. Despite this fact, the statistical structure of binaural sounds encountered in the natural environment and its dependence on the auditory scene have not yet been studied. In this paper we address this shortage. We performed binaural recordings of three real-world auditory scenes characterized by different acoustic properties and spatial dynamics. In the next step we extracted binaural cues - IPDs and ILDs and studied their marginal distributions by means of fitting parametric probability density functions. Parameters of fitted distributions allowed for an easy comparison of different scenes, and revealed which aspects change and which seem to remain invariant in different auditory environments. To analyze high-order statistics of binaural waveforms we performed Independent Component Analysis (ICA) of the signal, and studied properties of the learned features. The results obtained suggest how mechanisms of binaural hearing can be understood in terms of adaptation to natural stimulus statistics. They also allow for experimental predictions regarding neural computation and representation of the auditory space.
## Results
## Binaural spectra
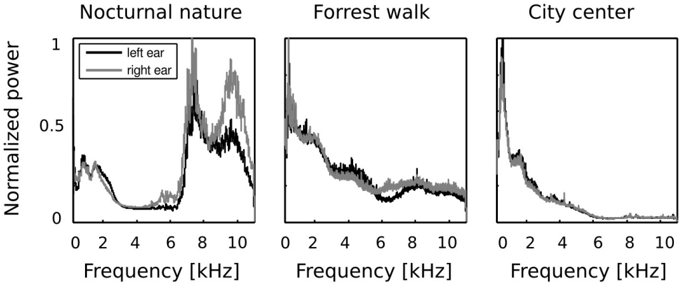
In the first step of the analysis, monaural Fourier spectra were compared with each other. Frequency spectra of recorded sounds are displayed on figure 2. Strong differences across all recorded auditory scenes were present. In two of them - the forrest walk scene and the city center scene, frequency spectrum had an exponential (power-law) shape, which is a characteristic signature of natural sounds [53]. Since the nocturnal nature scene was dominated by grasshoper sounds, its spectrum had two dominant peaks around 7 and 10 kHz. In all three cases, sounds in both ears contained a similar amount of energy in lower frequencies (below 4 kHz) - which is reflected by a good overlap of monaural spectra on the plots. In higher frequencies though, the spectral power was not always equally distributed in both ears. This difference is most strongly visible in the spectrum of the nocturnal nature scene. There, due to a persistent presence of a sound source (a grasshoper) closer to the right ear, corresponding frequencies were amplified with respect to the contralateral ear. Since the spatial configuration of the scene was static, this effect was not averaged out in time. Monaural spectra of the forrest walk scene overlapped to a much higher degree. A small notch in the left ear spectrum is visible around 6 kHz. This is most probably due to the fact that the recording subject stood next to a stream flowing at his right side for a period of time. The city center scene, has almost identical monaural spectra. This is a reflection of its rapidly changing spatial configuration - sound sources of similar quality (mostly human speakers) were present in all positions during the time of the recording.
## Interaural level difference statistics
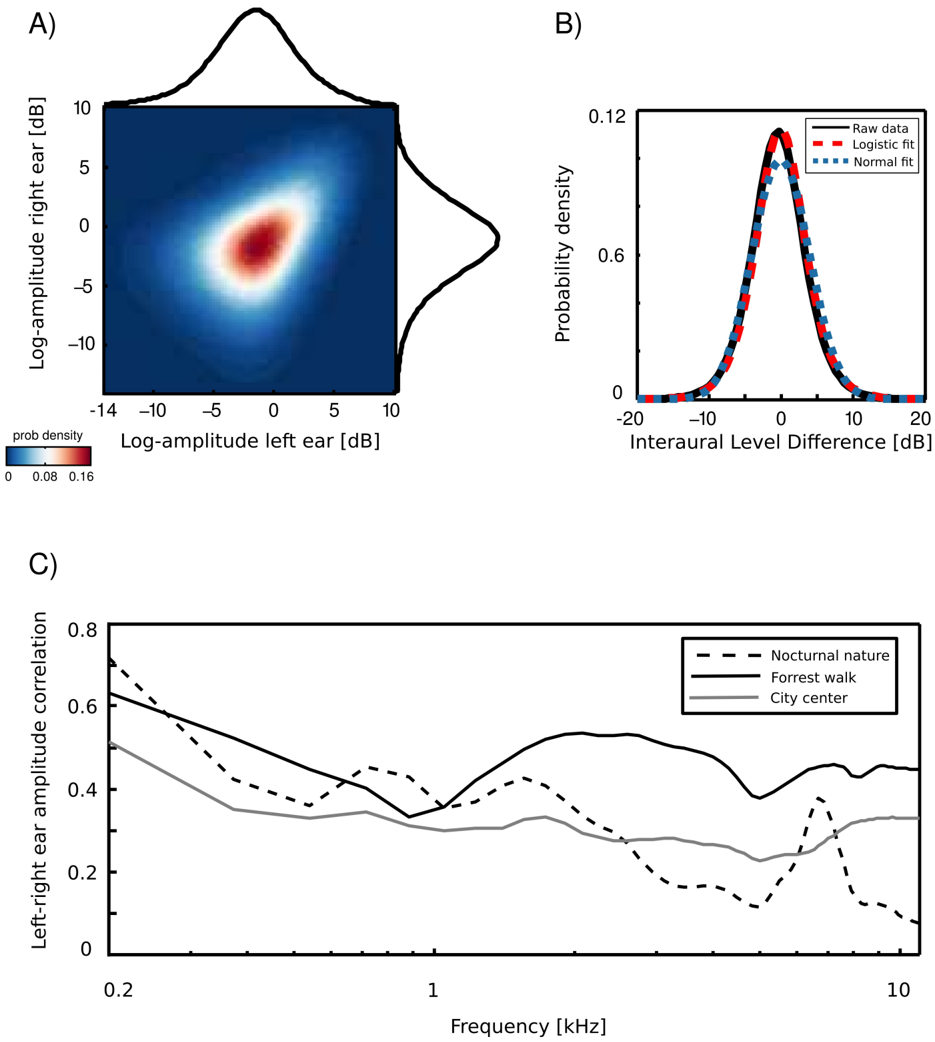
An example joint amplitude distribution in the left and the right ear is depicted in figure 3 A. It is not easily described by any parametric probability density function (pdf), however monaural amplitudes reveal a strong linear correlation. The correlation coefficient can be therefore used as a simple measure of interaural redundancy by indicating how similar the amplitude signal in both ears is, at a particular frequency channel. High correlation values would imply that both ears receive similar information, while low correlations indicate that the signal at both sides of the head is generated by different sources. Interaural amplitude correlations for all recorded
scenes are plotted as a function of frequency on figure 3 B. A general trend across the scenes is that correlations among low frequency channels (below 1 kHz) are strong (larger than 0 . 5) and decay with a frequency increase. Such a trend is expected due to the filtering properties of the head, which attenuates low frequencies much less than higher ones. The spatial structure of the scene also finds reflection in binaural correlation - for instance, a peak is visible in the nocturnal nature scene at 7 kHz. This is due to a presence of a spatially fixed source generating sound at this frequency (see figure 2). The most dynamic scene - city center - reveals, as expected, lowest correlations across most of the spectrum.
Interaural level differences ILD were computed separately in each frequency channel. Figure 3 C displays an example ILD distribution (black line) together with a best fitting Gaussian (blue dotted line) and logistic distribution (red dashed line). Logistic distributions provided the best fit to ILD distributions for all frequencies and recorded scenes, as confirmed by the KS-test (results not shown). ILD distribution at frequency ω was therefore defined as
$$\rho ( I L D _ { w } | u _ { w } , \sigma _ { w } ) = - \frac { e x p ( - I L D _ { w } ) } { a _ { w } ( 1 + e x p ( - I L ) } }$$
where µ ω and σ ω are frequency specific mean and scale parameters of the logistic pdf respectively. The variance of the logistic distribution is fully determined by the scale parameter.
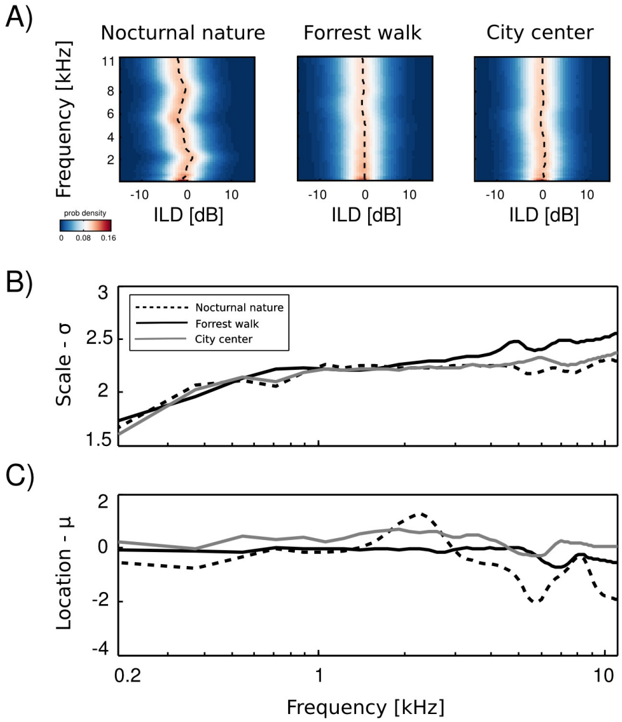
Empirical ILD distributions are plotted in figure 4 A. As can be immediately observed, they preserve similar shape in all frequency channels and auditory scenes, regardless of their type. The mean ( µ ω ) and scale ( σ ω ) parameters of the fitted distributions are plotted as a function of frequency in figures 4 B and C respectively. The mean of all distributions is very close to 0 dB in most cases. In the two non-static scenes, i.e., forrest walk and city center, deviations from 0 are very small. Marginal ILD distributions of the spatially constant scene - nocturnal nature - were slightly shifted away from zero for frequencies generated by a sound source of a fixed position. The scale parameter behaved differently than the mean. In all auditory scenes it grew monotonically with the increasing frequency. The increase was quite rapid for frequencies below 1 kHz - from 1 . 5 to 2. For higher frequencies the change was much smaller and in the 1 -11 kHz interval σ did not exceed the value of 2 . 5. What may be a surprising observation is the relatively small change in the ILD distribution, when comparing high and low frequencies. It is known that level differences become much more pronounced in the high frequency channels [30], and one could expect a strong difference with a frequency increase. These results can be partially explained by observing a close relationship between Fourier spectra of binaural sounds and means of ILD distributions. In a typical, natural setting sound sources on the left side of the head are qualitatively (spectrally) similar to the ones on the other side, therefore the spectral power in the same frequency bands remains similar in both ears. Average ILDs deviate from 0 if a sound source was present at a fixed position during the averaged time period. Increase in the ILD variance (defined by the scale parameter σ ) with increasing frequency, can be explained by the filtering properties of the head. While for lower frequencies a range of possible ILDs is low, since large spatial displacements generate weak ILD differences, in higher frequency regimes ILDs become more sensitive to the sound source position hence their variability grows. On the other hand, objects on both sides of the head reveal similar motion patterns and in this way reduce the ILD variability, which may account for the the small rate of change. Despite observed differences, ILD distributions revealed a strong invariance to frequency and were homogenous across different auditory scenes.
## Interaural phase difference statistics
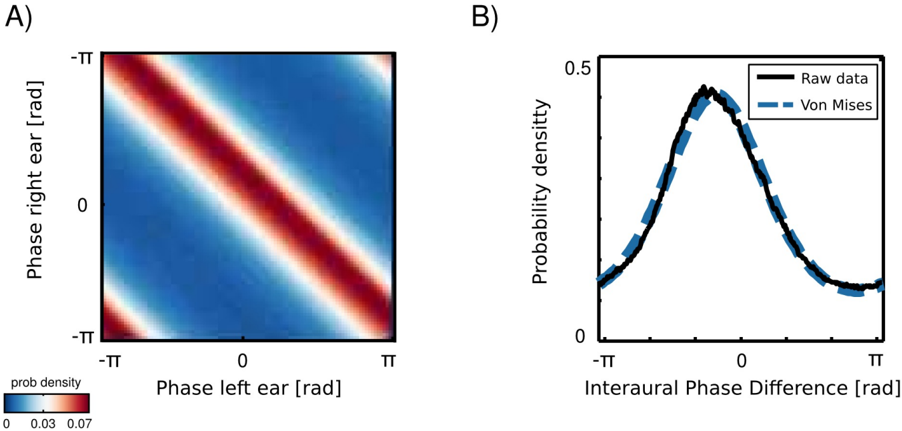
Marginal distributions of a univariate, monaural phases over a long time period are all uniform, since phase visits cyclically all values on a unit circle. An interesting structure appears in a joint distribution of left and right ear phase values from the same frequency channel (an example is plotted in figure 5). Monaural phases reveal dependence in their difference. This means that their joint probability is determined by the probability of their difference:
$$( 2 )$$
where φ L and φ R are instantenous phase values in the left and the right ear respectively. The well known physical mechanisms explain this effect. The sound wavefront reaches first the ear ipsilateral to the sound source and then, after a short delay the contralateral one. The temporal difference generates a phase offset, which is reflected in the joint distribution of monaural phases. This simple observation implies, however, that IPDs constitute an intrinsic statistical structure of the natural binaural signal.
IPD histograms were well approximated by the von Mises distribution (additional structure was present in IPDs from the forrest walk scene - see subsection ). A distribution of two monaural phase variables revealing dependence in the difference can be then written as a von Mises distribution of their differences:
$$\rho ( \phi _ { L , w } , \phi _ { R , w } ) = \rho ( I P D _ { w } | k _ { w } , \mu _ { w } ) = \frac { 2 m } { e ^ { - k \cos ( I P D _ { w } - \mu _ { w } ) }$$
where IPD ω = φ L,ω -φ R,ω is the IPD at frequency ω , µ ω and κ ω are frequency specific mean and concentration parameters and I 0 is the modified Bessel function of order 0. In such a case, the concentration parameter κ controls mutual dependence of monaural phases [12]. For large κ ω values φ L,ω and φ R,ω are strongly dependent and the dependence vanishes for κ = 0.
## IPD distributions
Figure 6 A depicts IPD histograms in all scenes depending on the frequency channel. Thick black lines mark IPD ω,max - the 'maximal IPD' value i.e. phase displacement corresponding to a time interval required for a sound to travel the entire interaural distance. IPD ω,max can be computed in a following way. Assuming a spherical head shape, the time period required by the sound wave to travel the distance between the ears is equal to:
$$\overrightarrow { I T D } = \frac { R _ { head } } { v s n d } ( \theta + \sin ( \theta ) )$$
where R head is the head radius, v snd the speed of sound and Θ the angular position of the sound source measured in radians from the midline. The ITD is maximized for sounds located directly oposite to one of the ears, deviating from the midline by π 2 (Θ = π 2 ). ITD max becomes
$$I T D _ { m a x } = \frac { R _ { h e a d } ( \pi + 1 ) . } { v s n d }$$
The maximal IPD is then computed separately in each frequency channel ω
$$1 P D _ { w , \max } = 2 π w I T D _ { \max . }$$
The above calculations assume a spherical head shape, which is a major simplification. It is, however, sufficient for the sake of the current analysis.
At low frequencies most IPD values do not exceed the 'forbidden' line, and the resulting plot has a triangular shape. This is a common tendency in IPD distributions, visible across all auditory scenes. Additionally, due to phase wrapping, for frequencies where π ≤ | IPD max | ≤ 2 π the probability mass is shifted away from the center of the unit circle towards the -π and π values, which is visible as blue, circular regions in the middle of the plot. This trend is not present in the forrest walk scene, where a clear peak at 0 radians is visible for almost all frequencies. This figure can be compared with figure 3 in [23] and 14 in [19]. The two panels below, i.e., figures 6 B and C, display plots of the κ and µ parameters of von Mises distributions as a function of frequency. The concentration parameter κ decreases in all three scenes from a value close to 1 . 5 (strong concentration) to below 0 . 5 in the 200 Hz to 500 Hz interval, which seems to be a robust property in all environments. Afterwards, small rebounds are visible. For auditory scenes recorded by a static subject, i.e., nocturnal nature and city center, rebounds occur at frequencies where IPD max corresponds to π multiplicities (this is again, an effect of phase wrapping). The κ value is higher for a more static scene - nocturnal nature - reflecting a lower IPD variance. For frequencies above 2 kHz, concentration converges to 0 in all three scenes. This means that IPD distributions become uniform and monaural phases mutually independent. The frequency dependence of the position parameter µ is visible on figure 6 C. For the forrest walk scene, IPD distributions were centered at the 0 value with an exception at 700 Hz. For the two scenes recorded by a static subject , distribution peaks were roughly aligned along the IPD max as long as it did not exceed -π or π value. In higher frequencies they varied much stronger, although one has to note that for distributions close to uniform ( κ → 0), position of the peak becomes an ill defined and arbitrary parameter.
Equations 4 - 6 allow to compute the 'maximal' IPD value ( IPD max ), constrained by the size of the organism's head. A single, point sound source in an anechoic environment would never generate IPD exceeding IPD max . In natural hearing conditions however, such IPDs occur due to the presence of two sound sources at both sides of the head or due to acoustic reflections [21]. Their presence is visible in figure 6 as probability mass lying outside of the black lines marking maximal IPD values at particular frequencies. Figure 7 displays a proportion of IPDs larger than the one defined by the head size plotted against frequency. The lines corresponding to three recorded auditory environments lie in parallel to each other, displaying almost the same trend up to a vertical shift. The highest proportion of IPDs exceeding the 'maximal' value was present in the nocturnal nature scene. This was most probably caused by a large number of very similar sound sources (grasshoppers) at each side of the head. They generated non-synchronized and strongly overlapping waveforms. Phase information in each ear resulted therefore from an acoustic summation of multiple sources, hence instantenous IPD was not directly related to a single source position and often exceeded the IPD max value. Surprisingly, IPDs in the most spatially dynamic scene - city center - did not exceed the IPD max limit as often. This may be due to a smaller number of sound sources present and may indicate that the proportion of 'forbidden' IPDs is a signature of a number of sound sources present in the scene. For nocturnal nature and city center scenes the proportion peaked at 400 Hz achieving values of 0 . 45 and 0 . 35 respectively. For a forrest walk scene, the peak at 400 Hz did not exceed the value of 0 . 31 at 200 Hz. All proportion curves converged to 0 at 734 Hz frequency, where IPD max = π .
## Separation of speech with single channel IPDs
As already mentioned before, IPD distributions at most frequency channels in the forrest walk scene revealed an additional property, namely a clear, sharp peak at 0 radians. This feature was not present in the two other, statically recorded scenes. As an example, IPD distribution at 561 Hz is depicted in figure 8 A. The histogram structure reflects the elevated presence of sounds
with IPDs close to 0 hence equal monaural phase values. Zero IPDs can be generated either by sources located at the midline (directly in front or directly in the back) or self-produced sounds such as speech, breathing or loud footsteps.
As visible in figure 8 two components contributed to the structure of the marginal IPD distribution - the sharp 'peak component' (dashed blue line) and the broad 'background' (dashed red line). Due to this property, IPD histograms were well suited to be modelled by a mixture model. This means that their pdf could be represented as a linear combination of two von Mises distributions in the following way
$$\sum _ { i = 1 } ^ { n } p ( C _ { i } ) p ( I P D _ { w _ { i } } | w _ { i } , p _ { w _ { i } } )$$
where κ ω ∈ R 2 and µ ω ∈ R 2 are parameter vectors, C i ∈ { 1 , 2 } are class labels, p ( C i ) are prior probabilities of class membership and p ( IPD ω | κ ω,i , µ ω,i ) are von Mises distributions defined by equation 3. A fitted mixture of von Mises distributions is also visible in figure 8 A, where dashed lines are mixture components and a continuous black line is the marginal distribution. It is clearly visible that a two-component mixture fits the data much better than a plain von Mises distribution. There is also an additional advantage of fitting such a mixture model, namely it allows to perform a classification problem and assign each IPD sample (and therefore each associated sound sample) to one of the two classes defined by mixture components. Since the prior over class labels is assumed to be uniform, this procedure is equivalent to finding a maximumlikelihood estimate ˆ C of C
$$c = \arg \max _ { c } p ( I P D _ { c } | C )$$
In this way, if no sound source at the midline is present, a separation of self generated sounds from the background should be easily performed using information from a single frequency channel. Results of a self-generated speech separation task are displayed in figure 8 B. A two-second binaural sound chunk included two self-spoken words with a background consisting of a flowing stream. Each sample was classified basing on an associated IPD value at 561 Hz. Samples belonging to the second, sharp component are coloured blue and background ones are red. It can be observed that the algorithm has successfully separated spoken words from the environmental noise. Audio samples are available in the supplementary material.
## Independent components of binaural waveforms
In this section, instead of studying predetermined features of the stimulus (binaural cues), we use binaural waveforms to train Independent Compnent Analysis (ICA) - a statistical model which optimizes a general-purpose objective - coding efficiency [8]. In the ICA model, short (8 . 7 ms) epochs of binaural sounds are assumed to be a linear superposition of basis functions multiplied by linear coefficients s (see figure 9 A). Linear coefficients are assumed to be independent and sparse , i.e., close to 0 for most of data samples in the training dataset. Basis functions learned by ICA can be interpreted as patterns of correlated variability present in the dataset.
Figure 9 B depicts exemplary basis functions learned from each recording. Each feature consists of two parts, representing signal in the left and the right ear (black and red colours respectively). Features trained on different recordings vary in their shape. Those differences are explicitely visible in spectrotemporal representations of basis functions depicted on figure 10. Each shape corresponds to an equiprobability contour of a Wigner distribution associated with a single basis function. Wigner distributions localize energy of a temporal signal in the time frequency plane. Left and right ear parts belonging to the same feature are plotted with
the same color. The obtained time-frequency tilings reveal a strong dependence on the auditory scene. Firstly basis function shapes are different - from time extended and frequency-localized in the city center scene, to temporally brief, instantenous features of the forrest walk scene. Despite shape differences, in each case, basis functions tile the time-frequency plane uniformly. Their shapes constitute an interesting aspect of the auditory scene and can be compared with results obtained by [1, 32]. This is, however not the focus of the current work.
Sounds of the most spatially static scene - nocturnal nature - were modelled mostly by features of the same spectrotemporal properties in each ear (with an anomally which occurred around 3 . 5 kHz). This is visible in figure 10 - blobs of the same color lie mostly in the same region on the left and the right ear plots. In more dynamic scenes, independent components (ICs) captured different, non-trivial dependencies. Pure frequency features learned from the city center recording had similar monaural parts below 3 . 5 kHz. Above this threshold, a cross-frequency interaural coupling appeared - in the right ear panel, blue colored features lie in the high frequency regime, while in the left ear they occupy a low frequency region. This means that to represent natural binaural signal efficiently, monaural information from different frequencies should be processed simultaneously. Interaural dependencies represented by ICs of the forrest walk scene were even more complex. Since most of the basis functions were much more temporal than spectral, time dependencies were also captured in addition to the spectral ones. High frequency events in the right ear were coupled with more temporally extended, low-frequency features of the left ear. Interestingly, tiling of the time-frequency plane associated with the right ear was not as uniform as for the left one.
The majority of learned basis functions was highly localized in frequency, which agrees with results obtained by [1, 32, 48]. However, some basis functions did not have well localized spectra. They were excluded from the analysis, that is why the number of basis functions varies across the analyzed auditory scenes. See materials and methods for the detailed discussion. To understand how spectral power was distributed in monaural parts of ICs, we computed a peak power ratio (PPR):
$$P P R = 1 0 \log _ { 1 0 } ( \frac { A _ { max , L } } { A _ { max , R } } )$$
where A max,L , A max,R are maximal spectrum values of the left and right ear parts of each IC respectively. Each circle in figure 11 represents a single IC. Its vertical and horizontal coordinates are monaural peak frequencies and colors encode the PPR value. Features which lie along the diagonal can be considered as a representation of 'classical' ILDs, since they encode features of the same frequency in each ear and differ only in level. ICs lying away from the diagonal with high absolute PPR values represent more monaural information, and those with the low absoulte PPR other aspects of the stimulus, such as interaural cross-frequency couplings. Figure 12 depicts proportion of features of same monaural frequencies (on diagonal) and those which bind different frequency channels (off diagonal). A pronounced difference among auditory scenes is visible in figure 11. The majority of basis functions learned from the nocturnal nature scene (161) clusters closely to the diagonal. The basis function set trained on the mostly dynamic scene (city center) separates into three clear subpopulations. Two of them, including 140 features were monaural. Monaural basis functions were dominated mostly by the spectrum of a single ear part, and the part representing the contralateral ear was of a very low frequency, close to a DC component. The binaural subpopulation contained 111 basis functions perfectly aligned with the diagonal. Such separation suggests that waveforms in both ears were highly independent and should be modelled using a large set of separate, monaural events. ICA trained on the forrest walk scene yielded a set of basis functions which was a compromise between nocturnal nature and city center scenes. Even though the highest number of features - 165 lied off the diagonal, the separation was not as sharp as for the city center scene. What clearly appeared was a division
into two subpopulations, members of which were dominated by the spectrum of one of the ears. ICs mostly coupled low frequencies ( < 2 kHz) from one ear with a broad range of frequencies in the other. Those properties may imply that in the case of this scene, both - features modelling binaural dependendencies and capturing purely monaural events - were required to model the data. To allow further comparison of learned ICs and known coding mechanisms in the binaural auditory system, we computed ILD and IPD cue values. This was done only for features encoding the same frequency information in both ears, since phase differences are ill defined otherwise, and auditory brainstem extracts cues mostly from the same frequency channels [21]. Results are visible in figure 13. IPDs represented by independent components separated into two channels in the city center and forrest walk scenes. The range of IPDs was higher for a more spatially varying scene, which is visible as a strong scatter of points.
For the nocturnal nature scene no such separation is visible. This is perhaps due to the fact that object positions were mostly fixed, generating lowly-varying IPDs captured by the learned ICs. Therefore the model did not have to generalize over a broader range of IPDs. ILDs in turn, in all scenes were separated into two distinct channels. The separation strength correlated with the scene's spatial variability and was highest for the city center scene and lowest for the nocturnal nature. Interestingly, in the latter one, ILD features were present also in high frequencies which was not the case in the two others. Also here, the separation of features seems to reflect the spatial structure and dynamics of the auditory scene.
## Discussion
Binaural cues are usually studied in a relationship to the angular position of the generating stimulus [18, 17, 24]. In probabilistic terms this corresponds to modelling the conditional probability distribution p ( cue | θ ), where θ is the angular stimulus location. According to the Bayes theorem, position inference given the cue can be then performed by: (a) computing the posterior distribution p ( θ | cue ) and (b) identifying θ for instance, as a maximum of the posterior distribution. Formally this process can be described by the following equations:
$$p ( \beta | cue ) \times p ( cue | \beta ) p ( cue )$$
$$\theta = \arg \max _ { q } p ( | c u e )$$
where ˆ θ is the estimated position. The Bayesian approach to sound localization has been succesfuly applied before, for instance to predict behavior and neural representation of binaural cues in the barn owl [18].
In the present study, we focused on marginal distributions of cues and binaural waveforms. This approach allows us to understand aspects of binaural hearing in the natural environment which are not directly related to the sound localization task. Marginal distributions p ( cue ) describe global properties of the stimulus to which the nervous system is exposed under natural conditions. Knowledge of a typical stimulus structure allows to predict properties of the sensory neurons [46, 5] and helps in understanding the complexity of the task such as binaural auditory scene analysis, when performed in ecological conditions.
## Binaural cues in complex auditory environments
Binaural scenes recorded and studied in this paper were selected to represent broad groups of possible auditory environments of different acoustic and spatial properties. In all three cases,
waveforms in each ear were for most of the time an acoustic summation of multiple sound sources. Additional factors, which influenced monaural stimuli were motion trajectories of objects and the listener, as well as sound reflections. Instantenous binaural cue values were therefore not generated by a single, point source, but were a function of a complex auditory scene. Inversion of a cue value to a sound position becomes, in such a setting, an inverse problem, since multiple scene configurations could give rise to the same cue value (for instance an ILD equal to 0 can be generated by a single source located at the midline, or two identical sources symmetricaly located on both sides of the head, see section ). In such scenarios, the sound localization task can not be performed as a simple inversion of a cue value to the sound position (the most simple case described by equations 10 and 11). It rather becomes equivalent to the cocktail party problem [36]. Localization of a sound source in complex listening situations has been a subject of substantial psychophysical [9] and electrophysiological [51, 29, 15, 6] research. An interesting theoretical model has been suggested by Faller and Merimaa [16]. The authors of this study proposed that to localize one sound source out of many present, the auditory system could use instantenous binaural cues only in time intervals when the left and the right ear waveforms are highly coherent (i.e. their cross-correlation peak exceeds a certain threshold). In such brief moments, ILD and IPD values would correspond to only a single source. This mechanism is able to explain numerous psychophysical studies. Meffin and Grothe [38] hypothesized that the auditory brainstem may perform low-pass filtering of localization cues to reject rapidly fluctuating 'spurious' cue values, which may originate from multiple sources. The aforemetioned mechanisms, however, involve a rejection of a large amount of information, by discarding 'ambiguous cues', which may still contain information useful in the auditory scene parsing. In very general terms, a useful strategy for the auditory system would be to use higher dimensional stimulus features (such as temporal cue sequences, or cross-frequency cue dependencies) to separate a source (or sources) of interest from the background and infere its spatial configuration. It has been demonstrated, for instance, that neurons in the Inferior Colliculus of the rat show a stronger response to dynamic, 'ecologically valid' IPD sequences, than to constant IPDs [49, 50]. In the auditory cortex of macaque monkeys, neurons become sensitive to even more complex IPD sequences [34]. Such properties may be examples of tuning to high-dimensional, binaural stimulus aspects. In the above mentioned view, instantenous binaural cues, as extracted by the early brainstem nuclei LSO and MSO [21], provide information useful in the auditory scene analysis task. In natural conditions however, their mere identification is not necessarily equivalent to the localization of the sound position. Binaural cues may rather serve as inputs to further computations (which are not necesserily limited to sound localization per se) performed in the higher stages of the binaural auditory pathway.
## Implications for neural processing and representation of binaural sounds
As predicted by physics of sound propagation, monaural phase values in natural environments reveal dependence in their difference. The strength of the dependence is measured by κ - the concentration parameter of the von Mises IPD distribution [12]. Interestingly, humans stop using IPDs to localize sounds above 1 . 5 kHz [54] i.e. the frequency regime, where monaural phases become marginally independent (as reflected by the decay of the κ parameter).
In anechoic environments, point sources of sound generate ITD values which are constrained by the head size of the listener. It has been, however, observed that in many species, IPD sensitive neurons have peaks of their tuning curves located outside of this 'physiological' range [21]. This representational strategy has been explained by suggesting that in mammals IPDs are encoded by the activity of two separate, broadly tuned neural channels. Notably, such a representation emerges as a consequence of maximizing Fisher information about naturally occuring IPDs [23].
Here, we demonstrate that in natural hearing conditions a substantial amount of IPDs (up to 45%) lies outside of the physiological range. Those IPD values may be a result of a reflection [20] or a presence of multiple spatially separate desynchronized sound sources [21]. Sound reflections generate reproducible cues and carry information about the spatial properties of the scene [20]. If a large IPD did not arise as a result of a reflection, it means that at least two sound sources contribute to the stimulus at the same frequency. Especially in the latter case, IPDs provide not only spatial information useful to identify the position of the sound, but become a strong source separation cue. Proportion of IPDs exceeding the physiological range decreased with growing frequency (since the maximal IPD limit increases). This observation agrees with the experimental data showing that in many species, neurons with low best frequency are tuned to large IPDs which often exceed the physiological range [35, 10, 22, 31]. Taken together, IPDs larger than predicted by the head size occur frequently in natural hearing conditions and carry important information. This can be an additional factor, explaining why in mammals, peaks of the IPD tuning curves lie outside of the physiological range. As demonstrated in section , interaural phase differences can be used for not explicitely spatial hearing tasks, such as extraction of self-generated speech (and potentially other sounds, such as steps). If there is no sound source present at the midline location (corresponding to 0 IPD), a simple classification procedure suffices to identify and separate vocalization of oneself from the background sound using information from a single frequency channel. Differentiation between self generated sounds and sounds of the environment is a behaviorally relevant task which has to be routinely performed by animals.
According to the Duplex Theory, ILDs contribute mostly to localization of high frequency sounds, since the head attenuates higher frequencies much stronger than lower ones [9]. Analysis of human Head Related Transfer Functions (HRTFs) shows that ILDs are almost constant at different spatial positions for low frequencies and become more variable (hence more informative) when frequency increases above 4 kHz [30]. For single sound sources in anechoic environments, ILDs can have values as large as 40 dB [30]. Based on those observations, one could expect that natural ILD distributions are strongly frequency dependent. Somewhat surprisingly, natural distributions reveal a quite homogenous structure across different frequency channels, which is well captured by the logistic distribution. Overall, averages are equal (or very close to) 0 dB in different auditory environments, and for all studied frequencies. Variance slightly increases with increasing frequency. Homogeneity of distribution forms and averages can be explained in the following way. Typical, natural auditory scenes consist of similar sound sources at both sides of the head (human speakers, grasshoppers, wind, etc). Each of the sound sources has a similar spectrum, hence they all contribute to waveforms in the left and right ear mutually cancelling each other. For this reason, ILD averages are close to 0 and have a similar shape in different environments. The variance increase can be explained by properties of the head related filtering since small movements of high frequency sources give rise to a large ILD variability. As mentioned before, interaural level differences are mostly believed to contribute to localization of high frequency sounds [9] since then they are large enough to be easily detectable. It has been, however, demonstrated that sound sources proximal to the listener can generate pronounced ILDs also in low frequencies (below 1 . 5 kHz) [11, 45]. Our results show that in natural environments the auditory system is exposed to a similar ILD distribution across all frequencies, including the low ones. The distribution includes also relatively large values (above 10 dB). Close sound sources and other environmental factors such as the wind perceived in only one ear generate large low-frequency ILDs. One could therefore speculate that neurons with low best frequencies should also form an ILD representation. Indeed, such neurons have been found in the Lateral Superior Olive (LSO) of the cat [52].
To go beyond studying one-dimensional features of the binaural signal (ILDs and IPDs), the probability distribution of short binaural waveforms was modelled by performing Independent
Component Analysis. A similar analysis in the visual domain was performed by Hoyer and Hyv¨ arinen [25] for binocular image pairs. The ICA algorithm has identified complex patterns of dependency, different for each studied auditory scene. Interestingly, the spectrotemporal shape of the monaural parts of the basis functions varied strongly across recorded auditory scenes. The obtained results can be compared with other studies which applied Independent Component Analysis to natural sounds [32, 1, 7]. Linear codes learned by the ICA model show that one should adopt different representations depending on the properties of the acoustic environment. A static scene (nocturnal nature) generated monaural waveforms, which were highly redundant, since the signal in each ear was originating mostly from the same source. For this reason, the majority of basis functions represented amplitude fluctuations in both ears and in the same frequency channels. When sound sources moved rapidly and independently from each other at both sides of the head, waveforms in each ear were much less redundant. That is why a representation of the dynamic binaural scene (city center) consisted of three, clearly separate populations of basis functions - two representing monaural signal, and one binaural. Interestingly, binaural functions coupled monaural channels of the same frequency. The moderatly dynamic scene (forrest walk) was best represented by basis functions which were mostly monaural and modelled a broad range of binaural cross-frequency dependencies. A variety of different dependency forms were captured, including temporal, spectral and spectrotemporal ones. This implies that information present in the binaural signal goes beyond instantenous binaural cue values. This notion goes in line with studies, which have found and characterized spectrotemporal binaural neurons at the higher stages of the auditory pathway [41, 39]. Binaural hearing in the natural environment may also rely on comparison of spectrotemporal information at both sides of the head.
## Conclusions
In the present study, we analyzed marginal statistics of binaural cues and waveforms. Thereby, we provided a general statistical characterization of the stimulus processed by the binaural auditory system in natural listening conditions. We have also made availible natural binaural recordings, which may be used by other researchers in the field. In a broad perspective, this study contributes to the lines of research that attempt to explain properties of the auditory system by analyzing natural stimulus structures. Further understanding of binaural hearing mechanisms will require a more systematic analysis of higher order stimulus statistics. This is the subject of future research.
## Materials and Methods
## Recorded scenes
The main goal of the study was to analyze cue distributions in different auditory environments. To this end, three auditory scenes of different spatial dynamics and acoustic properties were recorded. Each of the recordings lasted 12 minutes.
1. Nocturnal nature -the recording subject sat in a randomly selected position in the garden during a summer evening. During the recording the subject kept his head still, looking ahead, with his chin parallel to the ground. The dominating background sound are grasshopper calls. Other acoustic events included sounds of a distant storm and a few cars passing by on a near-by road. The spatial configuration of this scene did not change much in time - it was almost static.
2. City center - the recording subject sat in a touristic area of an old part of town, fixating the head as in the previous case. During the recording many moving and static human speakers were present. Contrasted with the previous example, the spatial configuration of the scene varied continuously.
3. Forrest walk - this recording was performed by a subject freely moving in the wooded area. A second speaker was present, engaged in a free conversation with the recording subject. In addition to speech, this scene included environmental sounds such as flowing water, cracks of broken sticks, leave crunching, wind etc. The binaural signal was affected not only by the spatial scene configuration, but also by the head and body motion patterns of the recording subject.
Two of the analyzed auditory scenes (nocturnal nature and city center) were recorded by a non-moving subject, therefore sound statistics were unaffected by the listener's motion patterns and self generated sounds. In the third scene (forrest walk) the subject was moving freely and speaking sparsely. Scene recordings are available in the supplementary material.
## Binaural recordings
Recordings were performed using Soundman OKM-II binaural microphones which were placed in the left and the right ear channels of the recording subject. A Soundman DR2 recorder was used to simultaneously record sound in both channels in an uncompressed wave format at 44100 Hz sampling rate. The head circumference of the recording subject was equal to 60 cm. Assuming a spherical head model this corresponds to a 9 . 5 cm head radius.
## Frequency filtering and cue extraction
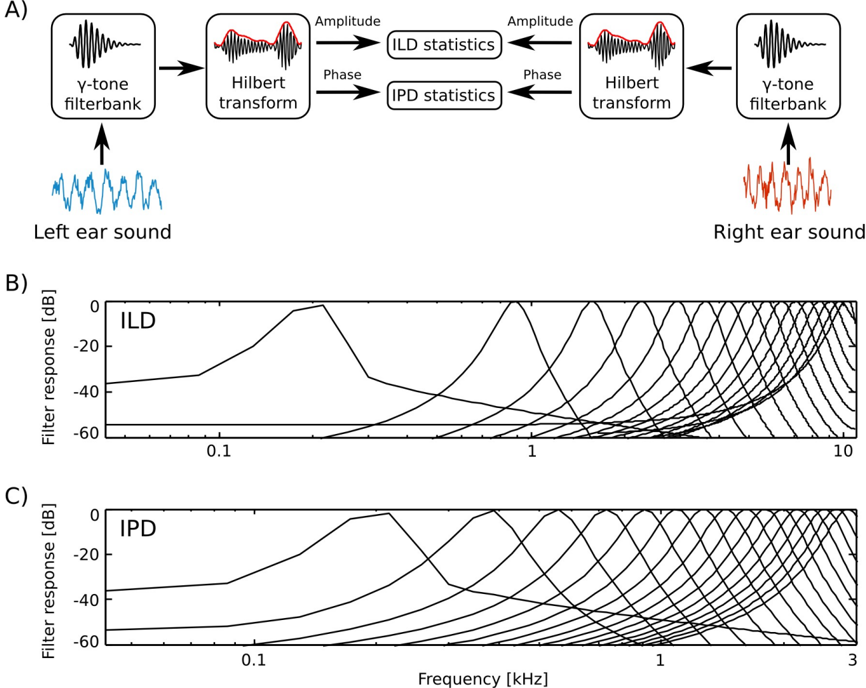
Prior to the analysis, raw recordings were down-sampled to the 22050 Hz sampling rate. The filtering and cue extraction pipeline is schematically depicted in figure 1
To emulate spectral decomposition of the signal performed by the cochlea, sound waveforms from each ear were transformed using a filterbank of 64 linear gammatone filters. Filter center frequencies were lineary spaced between 200 and 3000 Hz for IPD analysis and 200 and 10000 Hz for ILD analysis.
A Hilbert transform in each frequency channel was performed. In result, instantenous phase φ L,R ( ω, t ) and amplitude A L,R ( ω, t ) were extracted, separating level and phase information. Instantenous binaural cue values were computed in corresponding frequency channels ω from both ears according to the following equations:
$$\frac { I L D ( w , t ) } { I R ( w , t ) } = 1 0 \times \log _ { 1 0 } \frac { A _ { R } ( w , t ) } { A _ { L } ( w , t ) }$$
$$IP D ( w , t ) = \phi _ { L } ( w , t ) - \phi _ { R } ( w , t )$$
IPDs with an absolute value exceeding Π were wrapped to a [ -Π , Π] interval. Time series of IPD and ILD cues obtained in this way in each frequency channel were subjected to the further analysis.
## Independent Component Analysis
Independent Component Analysis (ICA) is a family of algorithms which attempt to find a linear transformation of the data that minimizes redundancy [28]. Given the data matrix X ∈ R n × m
(where n is the number of data dimensions and m number of samples), ICA finds a filter matrix W ∈ R n × n with
$$\vert x \vert = s$$
where the columns of X are data vectors x ∈ R n , the rows of W are linear filters w ∈ R n and S ∈ R n × m is a matrix of latent coefficients, which according to the assumptions are marginally independent. Equivalently the model can be defined using a basis function matrix A = W -1 , such that:
$$x = A S$$
The columns a ∈ R n of the matrix A are called basis functions. In modelling of neural systems they are usually interpreted as linear receptive fields forming an efficient code of the training data ensemble [28]. Each data vector can be represented as a linear combination of basis functions a , multiplied by linear coefficients s according to the equation 16.
$$x ( t ) = \sum _ { i } s _ { i } a _ { i } ( t )$$
where t indexes the data dimensions. The set of basis functions a is called a dictionary. ICA attempts to learn a linear, maximally non-redundant code, hence the latent coefficients s are assumed to be statistically independent i.e.
$$p ( s ) = \sum _ { i = 1 } ^ { n } p ( s _ { i } )$$
The marginal probability distributions p ( s i ) are typically assumed to be sparse (i.e. of high kurtosis), since natural sounds and images have an intrinsically sparse structure [40] and can be represented as a combination of a small number of primitives. In the current work we assumed a logistic distribution of the form:
$$p ( s _ { 1 } \vert u _ { 2 } s ) = \frac { e ^ { - s _ { 1 } } } { \{ 1 + e ^ { - s _ { 2 } } \} }$$
with position µ = 0 and the scale parameter ξ = 1. Basis functions were learned by maximizing the log-likelihood of the model via gradient ascent [28].
Prior to ICA learning, the recordings were downsampled to a 14700 Hz sampling rate (to obtain easy comparison with results in [32]). A training dataset was created by randomly drawing 100000 intervals each 128 samples long (corresponding to 8 . 7 ms).
## Acknowledgments
This work was funded by the DFG graduate college InterNeuro.
## References
- [1] Samer A Abdallah and Mark D Plumbley. If the independent components of natural images are edges, what are the independent components of natural sounds. In Proceedings of International Conference on Independent Component Analysis and Signal Separation (ICA2001) , pages 534-539, 2001.
- [2] H Attias and CE Schreiner. Temporal low-order statistics of natural sounds. Advances in neural information processing systems , pages 27-33, 1997.
- [3] Fred Attneave. Some informational aspects of visual perception. Psychological review , 61(3):183, 1954.
- [4] Horace B Barlow. Possible principles underlying the transformation of sensory messages. Sensory communication , pages 217-234, 1961.
- [5] Horace B Barlow. Unsupervised learning. Neural computation , 1(3):295-311, 1989.
- [6] Caitlin S Baxter, Brian S Nelson, and Terry T Takahashi. The role of envelope shape in the localization of multiple sound sources and echoes in the barn owl. Journal of neurophysiology , 109(4):924-931, 2013.
- [7] Anthony J Bell and Terrence J Sejnowski. Learning the higher-order structure of a natural sound*. Network: Computation in Neural Systems , 7(2):261-266, 1996.
- [8] Anthony J Bell and Terrence J Sejnowski. The independent components of natural scenes are edge filters. Vision research , 37(23):3327-3338, 1997.
- [9] Jens Blauert. Spatial hearing: the psychophysics of human sound localization . MIT press, 1997.
- [10] Antje Brand, Oliver Behrend, Torsten Marquardt, David McAlpine, and Benedikt Grothe. Precise inhibition is essential for microsecond interaural time difference coding. Nature , 417(6888):543-547, 2002.
- [11] Douglas S Brungart and William M Rabinowitz. Auditory localization of nearby sources. head-related transfer functions. The Journal of the Acoustical Society of America , 106:1465, 1999.
- [12] Charles F Cadieu and Kilian Koepsell. Phase coupling estimation from multivariate phase statistics. Neural computation , 22(12):3107-3126, 2010.
- [13] Nicole L Carlson, Vivienne L Ming, and Michael Robert DeWeese. Sparse codes for speech predict spectrotemporal receptive fields in the inferior colliculus. PLoS computational biology , 8(7):e1002594, 2012.
- [14] Johannes C Dahmen, Peter Keating, Fernando R Nodal, Andreas L Schulz, and Andrew J King. Adaptation to stimulus statistics in the perception and neural representation of auditory space. Neuron , 66(6):937-948, 2010.
- [15] Mitchell L Day, Kanthaiah Koka, and Bertrand Delgutte. Neural encoding of sound source location in the presence of a concurrent, spatially separated source. Journal of Neurophysiology , 108(9):2612-2628, 2012.
- [16] Christof Faller and Juha Merimaa. Source localization in complex listening situations: Selection of binaural cues based on interaural coherence. The Journal of the Acoustical Society of America , 116:3075, 2004.
- [17] Brian J Fischer. Optimal models of sound localization by barn owls. In Advances in Neural Information Processing Systems , pages 449-456, 2007.
- [18] Brian J Fischer and Jos´ e Luis Pe˜ na. Owl's behavior and neural representation predicted by bayesian inference. Nature neuroscience , 14(8):1061-1066, 2011.
- [19] Dan FM Goodman and Romain Brette. Spike-timing-based computation in sound localization. PLoS computational biology , 6(11):e1000993, 2010.
- [20] Boris Gour´ evitch and Romain Brette. The impact of early reflections on binaural cues. The Journal of the Acoustical Society of America , 132:9, 2012.
- [21] Benedikt Grothe, Michael Pecka, and David McAlpine. Mechanisms of sound localization in mammals. Physiological Reviews , 90(3):983-1012, 2010.
- [22] Kenneth E Hancock and Bertrand Delgutte. A physiologically based model of interaural time difference discrimination. The Journal of neuroscience , 24(32):7110-7117, 2004.
- [23] Nicol S Harper and David McAlpine. Optimal neural population coding of an auditory spatial cue. Nature , 430(7000):682-686, 2004.
- [24] Paul M Hofman and A John Van Opstal. Bayesian reconstruction of sound localization cues from responses to random spectra. Biological cybernetics , 86(4):305-316, 2002.
- [25] Patrik O Hoyer and Aapo Hyv¨ arinen. Independent component analysis applied to feature extraction from colour and stereo images. Network: Computation in Neural Systems , 11(3):191-210, 2000.
- [26] Anne Hsu, Sarah MN Woolley, Thane E Fremouw, and Fr´ ed´ eric E Theunissen. Modulation power and phase spectrum of natural sounds enhance neural encoding performed by single auditory neurons. The Journal of neuroscience , 24(41):9201-9211, 2004.
- [27] Aapo Hyv` earinen, Jarmo Hurri, and Patrick O Hoyer. Natural Image Statistics , volume 39. Springer, 2009.
- [28] Aapo Hyv` earinen, Jarmo Hurri, and Patrick O Hoyer. Natural Image Statistics , volume 39. Springer, 2009.
- [29] Clifford H Keller and Terry T Takahashi. Localization and identification of concurrent sounds in the owl's auditory space map. The Journal of neuroscience , 25(45):10446-10461, 2005.
- [30] Andrew J King, Jan WH Schnupp, and Timothy P Doubell. The shape of ears to come: dynamic coding of auditory space. Trends in cognitive sciences , 5(6):261-270, 2001.
- [31] Shigeyuki Kuwada and Tom C Yin. Binaural interaction in low-frequency neurons in inferior colliculus of the cat. i. effects of long interaural delays, intensity, and repetition rate on interaural delay function. Journal of Neurophysiology , 50(4):981-999, 1983.
- [32] Michael S Lewicki. Efficient coding of natural sounds. Nature neuroscience , 5(4):356-363, 2002.
- [33] Julia K Maier, Phillipp Hehrmann, Nicol S Harper, Georg M Klump, Daniel Pressnitzer, and David McAlpine. Adaptive coding is constrained to midline locations in a spatial listening task. Journal of Neurophysiology , 108(7):1856-1868, 2012.
- [34] Brian J Malone, Brian H Scott, and Malcolm N Semple. Context-dependent adaptive coding of interaural phase disparity in the auditory cortex of awake macaques. The Journal of neuroscience , 22(11):4625-4638, 2002.
- [35] David McAlpine, Dan Jiang, and Alan R Palmer. A neural code for low-frequency sound localization in mammals. Nature neuroscience , 4(4):396-401, 2001.
- [36] Josh H McDermott. The cocktail party problem. Current Biology , 19(22):R1024-R1027, 2009.
- [37] Josh H McDermott and Eero P Simoncelli. Sound texture perception via statistics of the auditory periphery: evidence from sound synthesis. Neuron , 71(5):926-940, 2011.
- [38] Hamish Meffin and Benedikt Grothe. Selective filtering to spurious localization cues in the mammalian auditory brainstem. The Journal of the Acoustical Society of America , 126:2437, 2009.
- [39] Lee M Miller, Monty A Escab´ ı, Heather L Read, and Christoph E Schreiner. Spectrotemporal receptive fields in the lemniscal auditory thalamus and cortex. Journal of Neurophysiology , 87(1):516-527, 2002.
- [40] Bruno A Olshausen and David J Field. Sparse coding with an overcomplete basis set: A strategy employed by v1? Vision research , 37(23):3311-3325, 1997.
- [41] Anqi Qiu, Christoph E Schreiner, and Monty A Escab´ ı. Gabor analysis of auditory midbrain receptive fields: spectro-temporal and binaural composition. Journal of Neurophysiology , 90(1):456-476, 2003.
- [42] Lord Rayleigh. On our perception of the direction of a source of sound. Proceedings of the Musical Association , 2:75-84, 1875.
- [43] F Rieke, DA Bodnar, and W Bialek. Naturalistic stimuli increase the rate and efficiency of information transmission by primary auditory afferents. Proceedings of the Royal Society of London. Series B: Biological Sciences , 262(1365):259-265, 1995.
- [44] Fred Rieke, David Warland, Rob Deruytervansteveninck, and William Bialek. Spikes: exploring the neural code (computational neuroscience). 1999.
- [45] Barbara G Shinn-Cunningham, Scott Santarelli, and Norbert Kopco. Tori of confusion: Binaural localization cues for sources within reach of a listener. The Journal of the Acoustical Society of America , 107:1627, 2000.
- [46] Eero P Simoncelli and Bruno A Olshausen. Natural image statistics and neural representation. Annual review of neuroscience , 24(1):1193-1216, 2001.
- [47] Nandini C Singh and Fr´ ed´ eric E Theunissen. Modulation spectra of natural sounds and ethological theories of auditory processing. The Journal of the Acoustical Society of America , 114:3394, 2003.
- [48] Evan C Smith and Michael S Lewicki. Efficient auditory coding. Nature , 439(7079):978-982, 2006.
- [49] Matthew W Spitzer and Malcolm N Semple. Interaural phase coding in auditory midbrain: influence of dynamic stimulus features. Science , 254(5032):721-724, 1991.
- [50] Matthew W Spitzer and Malcolm N Semple. Transformation of binaural response properties in the ascending auditory pathway: influence of time-varying interaural phase disparity. Journal of neurophysiology , 80(6):3062-3076, 1998.
- [51] Terry T Takahashi and Clifford H Keller. Representation of multiple sound sources in the owl's auditory space map. The Journal of neuroscience , 14(8):4780-4793, 1994.
- [52] Daniel J Tollin and Tom CT Yin. Interaural phase and level difference sensitivity in lowfrequency neurons in the lateral superior olive. The Journal of neuroscience , 25(46):1064810657, 2005.
- [53] Richard F Voss and John Clarke. 1/fnoise'in music and speech. Nature , 258:317-318, 1975.
- [54] Frederic L Wightman and Doris J Kistler. Sound localization. In Human psychophysics , pages 155-192. Springer, 1993.
## Figures
Figure 1: Preprocessing and cue extraction pipeline
<details>
<summary>Image 1 Details</summary>
### Visual Description
## Diagram: Auditory Signal Processing Pipeline and Frequency Response Analysis
### Overview
The image presents a technical diagram of a binaural auditory signal processing system, combining signal transformation workflows (Part A) with frequency response graphs for Interaural Level Differences (ILD) and Interaural Phase Differences (IPD) (Parts B and C). The system processes left/right ear sounds through filterbanks, Hilbert transforms, and statistical analysis to model spatial hearing cues.
---
### Components/Axes
#### Part A: Signal Processing Workflow
1. **Left Ear Path**:
- Input: "Left ear sound" (blue waveform)
- Components:
- `y-tone filterbank` → `Hilbert transform` → `Amplitude` → `ILD statistics`
- `Phase` → `IPD statistics`
2. **Right Ear Path**:
- Input: "Right ear sound" (red waveform)
- Components:
- `y-tone filterbank` → `Hilbert transform` → `Amplitude` → `ILD statistics`
- `Phase` → `IPD statistics`
3. **Key Elements**:
- Arrows indicate signal flow direction
- Red/blue waveforms distinguish left/right ear inputs
- Hilbert transform outputs are highlighted in red
#### Part B: ILD Frequency Response
- **Axes**:
- X-axis: Frequency (kHz), logarithmic scale (0.1 to 10 kHz)
- Y-axis: Filter response (dB), linear scale (-60 to 0 dB)
- **Legend**: No explicit legend; lines represent multiple frequency response curves
#### Part C: IPD Frequency Response
- **Axes**:
- X-axis: Frequency (kHz), logarithmic scale (0.1 to 3 kHz)
- Y-axis: Filter response (dB), linear scale (-60 to 0 dB)
- **Legend**: No explicit legend; lines represent multiple frequency response curves
---
### Detailed Analysis
#### Part A: Signal Processing Flow
1. **Left Ear**:
- Blue waveform enters `y-tone filterbank`, splitting into amplitude/phase components.
- Amplitude → `ILD statistics` (interaural level differences).
- Phase → `IPD statistics` (interaural phase differences).
2. **Right Ear**:
- Red waveform follows identical processing steps but with distinct Hilbert transform output.
- Amplitude/phase data from both ears are compared to compute ILD/IPD.
#### Part B: ILD Frequency Response
- **Trends**:
- Multiple curves show filter responses peaking at ~1 kHz (highest amplitude).
- Responses decline sharply above 10 kHz.
- Lower frequencies (<0.1 kHz) exhibit minimal filtering.
- **Notable Features**:
- Curves exhibit "notch" patterns at mid-frequencies (1–10 kHz), suggesting bandpass filtering.
#### Part C: IPD Frequency Response
- **Trends**:
- Peaks occur at ~0.1 kHz (low-frequency dominance).
- Responses diminish above 1 kHz, with near-zero filtering beyond 3 kHz.
- Mid-frequency ranges (0.5–2 kHz) show moderate filtering.
---
### Key Observations
1. **ILD vs. IPD Frequency Sensitivity**:
- ILD is most sensitive to mid/high frequencies (1–10 kHz).
- IPD is most sensitive to low frequencies (0.1–1 kHz).
2. **Symmetry in Processing**:
- Both ears use identical processing steps, emphasizing binaural comparison.
3. **Waveform Differences**:
- Left ear (blue) and right ear (red) waveforms differ in amplitude/phase, critical for spatial localization.
---
### Interpretation
This system models how humans localize sound sources using ILD and IPD. The frequency-dependent filtering in Parts B and C reflects the human auditory system's reliance on:
- **ILD** for high-frequency sound localization (e.g., speech consonants).
- **IPD** for low-frequency sound localization (e.g., vowel formants).
The Hilbert transform in Part A extracts instantaneous amplitude/phase data, enabling precise statistical analysis of interaural differences. The absence of a legend in Parts B/C suggests the curves represent a range of filter settings or experimental conditions, requiring further context for interpretation.
</details>
Figure 2: Frequency spectra of binaural recordings
<details>
<summary>Image 2 Details</summary>
### Visual Description
## Line Graphs: Normalized Power vs. Frequency Across Environments
### Overview
The image contains three line graphs comparing normalized power (y-axis) across frequency (x-axis) for three environments: "Nocturnal nature," "Forrest walk," and "City center." Each graph includes two data series: "left ear" (black line) and "right ear" (gray line). The graphs are positioned side-by-side, with legends in the top-left corner of each plot.
### Components/Axes
- **X-axis**: Frequency [kHz], ranging from 0 to 10 kHz in all graphs.
- **Y-axis**: Normalized power, scaled from 0 to 1.
- **Legends**:
- Top-left of each graph.
- Labels: "left ear" (black) and "right ear" (gray).
- **Graph Titles**:
- "Nocturnal nature" (leftmost graph).
- "Forrest walk" (middle graph).
- "City center" (rightmost graph).
### Detailed Analysis
#### Nocturnal nature
- **Left ear (black)**:
- Peaks at ~2.5 kHz (normalized power ~0.8) and ~7.5 kHz (normalized power ~0.7).
- Gradual decline after 8 kHz.
- **Right ear (gray)**:
- Peaks at ~2.5 kHz (normalized power ~0.6) and ~7.5 kHz (normalized power ~0.5).
- Slightly lower amplitude than left ear.
#### Forrest walk
- **Left ear (black)**:
- Gradual decline from ~0.6 at 0 kHz to ~0.2 at 10 kHz.
- No distinct peaks.
- **Right ear (gray)**:
- Similar trend to left ear but consistently ~0.1 lower across all frequencies.
#### City center
- **Left ear (black)**:
- Sharp drop at ~1 kHz (normalized power ~0.8 to ~0.2).
- Gradual decline to ~0.05 at 10 kHz.
- **Right ear (gray)**:
- Similar sharp drop at ~1 kHz (normalized power ~0.6 to ~0.1).
- Slightly lower amplitude than left ear.
### Key Observations
1. **Left ear dominance**: In all environments, the left ear consistently shows higher normalized power than the right ear.
2. **Frequency-specific patterns**:
- Nocturnal nature: Peaks at 2.5 kHz and 7.5 kHz suggest sensitivity to mid-to-high-frequency sounds.
- Forrest walk: Uniform decline indicates ambient noise reduction with increasing frequency.
- City center: Sharp drop at 1 kHz implies a dominant low-frequency noise source (e.g., traffic).
3. **Environmental contrast**:
- Nocturnal nature and Forrest walk show smoother trends, while City center exhibits abrupt changes.
### Interpretation
The data suggests that auditory sensitivity or exposure varies significantly across environments. The left ear’s consistent higher power across all graphs may indicate anatomical asymmetry or preferential sound localization. The sharp drop in the City center graph at 1 kHz aligns with urban noise profiles dominated by low-frequency sources (e.g., engines, machinery). The gradual declines in Forrest walk and Nocturnal nature graphs suggest natural environments have more balanced frequency distributions.
**Uncertainties**: Exact normalized power values are approximate due to the absence of gridlines or numerical annotations. Peak frequencies (e.g., 2.5 kHz) are estimated based on visual alignment with the x-axis.
</details>
Figure 3: Binaural amplitude statistics. A) An exemplary plot of joint amplitude distribution in both ears B) ILD distribution for a fixed channel toghether with a Gaussian and a logistic fit C) Interaural correlations of amplitudes across frequency channels
<details>
<summary>Image 3 Details</summary>
### Visual Description
## Heatmap and Line Graphs: Interaural Amplitude Correlation and Environmental Analysis
### Overview
The image contains three panels (A, B, C) analyzing interaural amplitude relationships and environmental correlations. Panel A is a heatmap of log-amplitude correlations between left and right ears. Panel B compares raw data with logistic and normal distribution fits for interaural level differences. Panel C shows frequency-dependent amplitude correlations across three environments.
### Components/Axes
**Panel A (Heatmap):**
- **X-axis**: Log-amplitude left ear [dB] (-14 to 10 dB)
- **Y-axis**: Log-amplitude right ear [dB] (-10 to 10 dB)
- **Color scale**: Probability density (0 to 0.16, blue to red)
- **Contour line**: Black outline tracing peak probability region
- **Legend**: Color bar labeled "prob density" with blue (low) to red (high)
**Panel B (Line Graph):**
- **X-axis**: Interaural Level Difference [dB] (-20 to 20 dB)
- **Y-axis**: Probability density (0 to 0.12)
- **Lines**:
- Black: Raw data
- Red dashed: Logistic fit
- Blue dotted: Normal fit
- **Legend**: Top-right corner
**Panel C (Line Graph):**
- **X-axis**: Frequency [kHz] (0.2 to 10 kHz)
- **Y-axis**: Left-right ear amplitude correlation (0 to 0.8)
- **Lines**:
- Dashed: Nocturnal nature
- Solid: Forrest walk
- Dotted: City center
- **Legend**: Top-right corner
### Detailed Analysis
**Panel A:**
- Peak probability density (0.16) occurs at ~0 dB for both ears, forming a circular region.
- Probability decreases radially outward, with 90% of data within ±5 dB of both ears.
- Contour line encloses 80% of the probability mass.
**Panel B:**
- Raw data (black) peaks at 0 dB with a sharp drop-off.
- Logistic fit (red) closely matches raw data, with 95% of data within ±10 dB.
- Normal fit (blue) underestimates tails, showing 10% lower probability beyond ±15 dB.
**Panel C:**
- **Nocturnal nature** (dashed): Starts at 0.7 correlation at 0.2 kHz, declines to 0.3 by 10 kHz.
- **Forrest walk** (solid): Peaks at 0.6 at 0.5 kHz, dips to 0.2 at 1 kHz, recovers to 0.4 at 10 kHz.
- **City center** (dotted): Flat at 0.3-0.4 across all frequencies.
### Key Observations
1. **Panel A**: Strongest correlation occurs when left and right ear amplitudes are nearly equal (0 dB difference).
2. **Panel B**: Logistic distribution better models the data than the normal distribution, particularly in the tails.
3. **Panel C**:
- Nocturnal environments show highest frequency-dependent correlation.
- Forrest walk exhibits a notable 1 kHz dip, possibly indicating resonance effects.
- City center maintains consistently lower correlations, suggesting ambient noise interference.
### Interpretation
The data demonstrates that interaural amplitude correlations are strongest when sound sources are equidistant from both ears (Panel A). The logistic fit's superiority over the normal distribution (Panel B) suggests environmental noise follows a skewed distribution rather than a Gaussian one.
Environmental context significantly impacts correlation strength (Panel C):
- **Nocturnal nature**'s declining correlation with frequency may reflect directional sound sources (e.g., rustling leaves).
- **Forrest walk**'s 1 kHz anomaly could indicate human-made noise (e.g., footsteps) resonating at that frequency.
- **City center**'s flat correlation implies pervasive broadband noise masking directional cues.
These findings highlight how spatial hearing varies with environmental acoustics, with potential applications in auditory prosthetics and noise-cancellation systems.
</details>
Figure 4: Interaural level difference distributions. A) Histograms plotted as a function of frequency B)
<details>
<summary>Image 4 Details</summary>
### Visual Description
## Heatmaps and Line Graphs: Environmental Sound Analysis
### Overview
The image presents three heatmaps (A) and two line graphs (B, C) analyzing sound characteristics across three environments: "Nocturnal nature," "Forrest walk," and "City center." The heatmaps visualize frequency-intensity relationships, while the line graphs track scale (σ) and location (μ) metrics across frequencies.
### Components/Axes
**A) Heatmaps**
- **X-axis**: ILD [dB] (Intensity Level Difference), ranging from -10 to +10 dB.
- **Y-axis**: Frequency [kHz], ranging from 2 to 11 kHz.
- **Color Scale**: Probability density (0 to 0.16), with blue (low) to red (high).
- **Legend**: Bottom-left, indicating "prob density" with a gradient bar.
- **Annotations**: Dashed vertical lines in each heatmap (position varies slightly).
**B) Line Graph (Scale - σ)**
- **X-axis**: Frequency [kHz], 0.2 to 10 kHz.
- **Y-axis**: Scale - σ (unitless, 1.5 to 3).
- **Lines**:
- Dashed: Nocturnal nature (peaks at ~1 kHz, ~2.5 σ).
- Solid: Forrest walk (steady increase to ~2.8 σ at 10 kHz).
- Gray: City center (flat ~2.5 σ, minor fluctuations).
- **Legend**: Top-right, matching line styles to labels.
**C) Line Graph (Location - μ)**
- **X-axis**: Frequency [kHz], 0.2 to 10 kHz.
- **Y-axis**: Location - μ (unitless, -4 to +2).
- **Lines**:
- Dashed: Nocturnal nature (oscillates between -1 and +1 μ).
- Solid: Forrest walk (stable near 0 μ).
- Gray: City center (peaks at ~1.5 μ at 5 kHz, dips to -2 μ at 10 kHz).
- **Legend**: Top-right, consistent with B.
### Detailed Analysis
**A) Heatmaps**
- **Nocturnal nature**: Vertical orange band centered at 0 dB ILD, broadening at 4–6 kHz.
- **Forrest walk**: Narrower orange band at 0 dB ILD, sharper at 3–5 kHz.
- **City center**: Broadest orange band at 0 dB ILD, extending to ±5 dB ILD.
**B) Scale - σ Trends**
- **Nocturnal nature**: Peaks at ~1 kHz (2.5 σ), then declines.
- **Forrest walk**: Gradual rise from 1.8 σ (0.2 kHz) to 2.8 σ (10 kHz).
- **City center**: Flat ~2.5 σ, minor dip at 3 kHz.
**C) Location - μ Trends**
- **Nocturnal nature**: Sinusoidal pattern (0.2–10 kHz: -1 to +1 μ).
- **Forrest walk**: Stable near 0 μ, slight dip at 5 kHz.
- **City center**: Peaks at 1.5 μ (5 kHz), drops to -2 μ (10 kHz).
### Key Observations
1. **Heatmaps**: City center shows broader ILD spread, suggesting higher frequency variability.
2. **Line Graphs**:
- **Scale (σ)**: Forrest walk exhibits the highest σ at high frequencies.
- **Location (μ)**: City center has the most pronounced μ fluctuations.
3. **Dashed Lines**: In heatmaps, align with frequency peaks in line graphs (e.g., 1 kHz in B).
### Interpretation
The data suggests environmental differences in sound propagation:
- **Nocturnal nature** and **Forrest walk** show localized frequency-intensity peaks, likely due to natural acoustic reflections.
- **City center** exhibits broader ILD distributions and μ variability, indicating urban noise complexity (e.g., reflections, reverberation).
- **Scale (σ)** trends imply that sound energy distribution is most variable in urban settings at high frequencies.
- **Location (μ)** anomalies in the city center may reflect interference patterns from dense infrastructure.
The dashed lines in heatmaps correlate with σ peaks in line graphs, suggesting a relationship between intensity distribution and scale metrics. Urban environments demonstrate greater acoustic heterogeneity compared to natural settings.
</details>
Figure 5: Binaural phase statistics A) Exemplary joint probability distribution of monaural phases B) An IPD histogram (black line) and a fitted von-Mises distribution
<details>
<summary>Image 5 Details</summary>
### Visual Description
## Heatmap and Line Graph: Phase Relationship Analysis
### Overview
The image contains two panels:
- **Panel A**: A heatmap showing probability density as a function of phase differences between left and right ears.
- **Panel B**: A line graph comparing raw data and a Von Mises distribution for interaural phase differences.
### Components/Axes
#### Panel A (Heatmap)
- **X-axis**: "Phase left ear [rad]" (range: -π to π, labeled as -TT to TT).
- **Y-axis**: "Phase right ear [rad]" (range: -π to π, labeled as -TT to TT).
- **Color Scale**: Probability density (0.03 to 0.07), with red indicating higher density.
- **Legend**: Located at the bottom-left corner, labeled "prob density" with a gradient from blue (low) to red (high).
#### Panel B (Line Graph)
- **X-axis**: "Interaural Phase Difference [rad]" (range: -π to π, labeled as -TT to TT).
- **Y-axis**: "Probability density" (range: 0 to 0.5).
- **Legend**: Located at the top-right corner, with two entries:
- **Black line**: "Raw data"
- **Blue dashed line**: "Von Mises"
### Detailed Analysis
#### Panel A
- The heatmap exhibits a **diagonal red stripe** from the bottom-left to top-right, indicating a strong correlation between the phases of the left and right ears.
- Probability density is highest along this diagonal (red regions) and decreases toward the edges (blue regions).
- The color gradient confirms that the maximum probability density (~0.07) occurs near the diagonal, while minimum density (~0.03) is at the corners.
#### Panel B
- The **raw data** (black line) forms a symmetric unimodal distribution centered at 0 rad, peaking at ~0.45 probability density.
- The **Von Mises distribution** (blue dashed line) closely follows the raw data, with a peak at 0 rad and a slight deviation in the tails.
- Both distributions are symmetric about 0 rad, with no significant outliers.
### Key Observations
1. **Panel A**: The diagonal red stripe suggests a linear relationship between the phases of the two ears, with maximum probability when their phases are equal (0 rad difference).
2. **Panel B**: The Von Mises distribution closely matches the raw data, indicating that the phase differences follow a circular von Mises distribution, typical for angular data.
3. **Color Consistency**: In Panel A, red regions align with the legend’s high-density values, and blue regions align with low-density values.
### Interpretation
- **Panel A** demonstrates that the phases of the left and right ears are strongly correlated, with the highest likelihood of matching phases (0 rad difference). This could reflect binaural hearing mechanisms or synchronization in auditory processing.
- **Panel B** validates that the raw data aligns with the theoretical Von Mises distribution, confirming that interaural phase differences are circularly symmetric and concentrated near 0 rad. The slight deviation in the tails may indicate minor noise or measurement variability.
- The use of the Von Mises distribution (a circular analog of the Gaussian distribution) is appropriate here, as phase data inherently wraps around 2π radians.
*Note: All values are approximate, with uncertainty arising from the visual representation of the heatmap and line graph.*
</details>
Figure 6: IPD distributions. A) Histograms B) Concentration parameter κ ω as a function of frequency C) Position parameter µ ω as a function of frequency
<details>
<summary>Image 6 Details</summary>
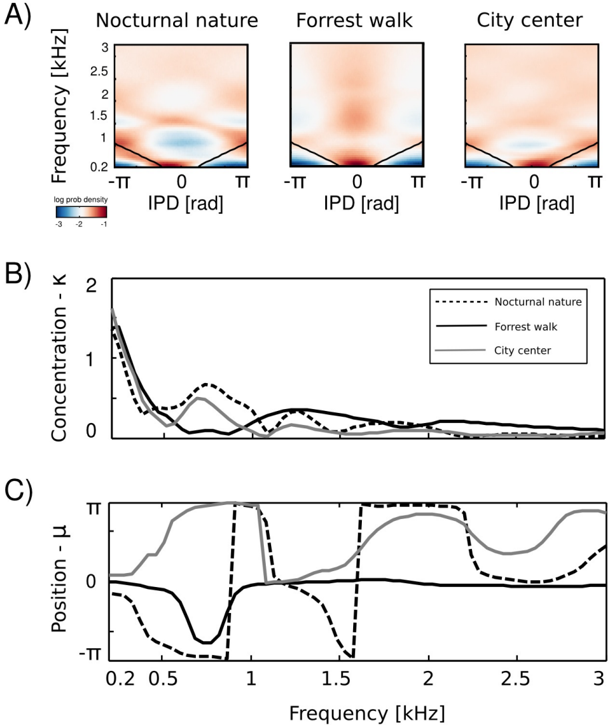
### Visual Description
## Heatmaps and Line Graphs: Frequency vs. IPD and Concentration/Position
### Overview
The image contains three visualizations comparing environmental data across three categories: "Nocturnal nature," "Forrest walk," and "City center." Part A shows heatmaps of frequency (kHz) vs. interaural phase difference (IPD, radians), while Parts B and C display line graphs of concentration (K) and position (μ) vs. frequency (kHz). All visualizations use color-coded lines and legends to differentiate categories.
---
### Components/Axes
#### Part A (Heatmaps)
- **X-axis**: IPD [rad] (labeled with -π, 0, π)
- **Y-axis**: Frequency [kHz] (0.2 to 3 kHz)
- **Color legend**: Log prob density (-3 to -1, blue to red gradient)
- **Legend placement**: Bottom-left corner
- **Categories**: Nocturnal nature, Forrest walk, City center (labeled at top)
#### Part B (Concentration vs. Frequency)
- **X-axis**: Frequency [kHz] (0.2 to 3 kHz)
- **Y-axis**: Concentration - K (0 to 2)
- **Lines**:
- Dashed: Nocturnal nature
- Solid: Forrest walk
- Gray: City center
- **Legend placement**: Top-right corner
#### Part C (Position vs. Frequency)
- **X-axis**: Frequency [kHz] (0.2 to 3 kHz)
- **Y-axis**: Position - μ (-π to π)
- **Lines**:
- Dashed: Nocturnal nature
- Solid: Forrest walk
- Gray: City center
- **Legend placement**: Top-right corner
---
### Detailed Analysis
#### Part A (Heatmaps)
- **Nocturnal nature**:
- High log prob density (red) near 0.2 kHz and -π/π IPD.
- Gradual decline in intensity toward higher frequencies.
- **Forrest walk**:
- Concentrated red region near 1.5 kHz and 0 IPD.
- Symmetric distribution around 0 IPD.
- **City center**:
- Broad red band centered at 1 kHz and 0 IPD.
- Lower intensity at extremes of IPD (±π).
#### Part B (Concentration vs. Frequency)
- **Nocturnal nature (dashed)**:
- Sharp peak at ~0.3 kHz (K ≈ 1.8).
- Rapid decline to ~0.1 kHz at 1.5 kHz.
- **Forrest walk (solid)**:
- Moderate peak at ~0.5 kHz (K ≈ 1.2).
- Gradual decline to ~0.05 kHz at 2.5 kHz.
- **City center (gray)**:
- Flat baseline (K ≈ 0.1) until ~1.2 kHz.
- Sharp rise to K ≈ 0.3 at 2.5 kHz.
#### Part C (Position vs. Frequency)
- **Nocturnal nature (dashed)**:
- Sharp drop from π to -π at ~0.3 kHz.
- Oscillatory behavior with minima at ~1.5 kHz.
- **Forrest walk (solid)**:
- Stable position near 0 until ~1 kHz.
- Sudden shift to -π at ~1.2 kHz.
- **City center (gray)**:
- Gradual oscillation between π and -π.
- Peaks at ~0.8 kHz and ~2.2 kHz.
---
### Key Observations
1. **Environmental differences**:
- City center shows higher frequency-dependent variability in concentration (Part B) and position (Part C).
- Forrest walk exhibits intermediate IPD sensitivity compared to nocturnal nature and city center.
2. **IPD-frequency relationships**:
- Nocturnal nature has strong IPD coupling at low frequencies (<1 kHz).
- City center shows broader IPD distribution across frequencies.
3. **Anomalies**:
- Forrest walk’s abrupt position shift at 1.2 kHz (Part C) suggests a threshold effect.
- City center’s late-frequency concentration spike (Part B) may indicate urban noise interference.
---
### Interpretation
The data suggests that environmental context significantly influences auditory processing metrics:
- **Nocturnal nature** prioritizes low-frequency IPD coupling, likely for spatial localization in quiet environments.
- **Forrest walk** balances IPD sensitivity and position stability, reflecting moderate acoustic complexity.
- **City center** demonstrates frequency-dependent position variability, consistent with broadband noise masking spatial cues.
The heatmaps (Part A) reveal that IPD sensitivity peaks at distinct frequencies for each environment, while the line graphs (Parts B-C) quantify how concentration and position metrics evolve with frequency. The abrupt shifts in Part C (Forrest walk) and Part B (City center) highlight critical frequency thresholds where environmental acoustics alter perceptual processing.
</details>
Figure 7: Proportion of IPDs exceeding the 'maximal IPD' threshold in each frequency channel
<details>
<summary>Image 7 Details</summary>
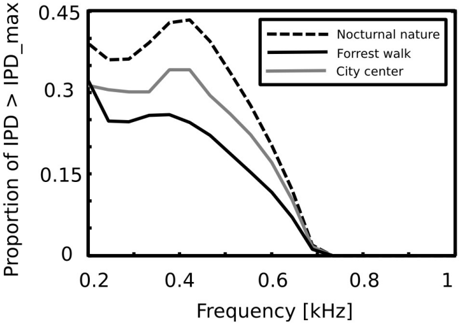
### Visual Description
## Line Graph: Proportion of IPD > IPD_max vs Frequency [kHz]
### Overview
The graph displays three frequency-dependent trends in the proportion of IPD (Interaural Phase Difference) exceeding a maximum threshold (IPD_max) across three environments: Nocturnal nature, Forrest walk, and City center. The y-axis represents the proportion (0–0.45), and the x-axis represents frequency (0.2–1 kHz). All lines converge near 0.8 kHz, suggesting a shared attenuation pattern at higher frequencies.
### Components/Axes
- **Y-axis**: "Proportion of IPD > IPD_max" (0 to 0.45, linear scale).
- **X-axis**: "Frequency [kHz]" (0.2 to 1, linear scale).
- **Legend**: Located in the top-right corner, with three entries:
- Dashed line: Nocturnal nature
- Solid black line: Forrest walk
- Solid gray line: City center
### Detailed Analysis
1. **Nocturnal nature (dashed line)**:
- Starts at ~0.3 at 0.2 kHz.
- Peaks at ~0.45 at ~0.4 kHz.
- Declines sharply to ~0.1 at ~0.6 kHz.
- Drops to near 0 by 0.8 kHz.
2. **Forrest walk (solid black line)**:
- Begins at ~0.25 at 0.2 kHz.
- Peaks at ~0.35 at ~0.3 kHz.
- Declines to ~0.15 at ~0.5 kHz.
- Reaches ~0.05 at 0.8 kHz.
3. **City center (solid gray line)**:
- Starts at ~0.3 at 0.2 kHz.
- Peaks at ~0.3 at ~0.35 kHz.
- Declines to ~0.1 at ~0.5 kHz.
- Drops to ~0.02 at 0.8 kHz.
### Key Observations
- **Nocturnal nature** exhibits the highest initial proportion and widest frequency range before attenuation.
- **Forrest walk** and **City center** show similar peak proportions (~0.35 and ~0.3, respectively) but differ in frequency of peak occurrence.
- All lines converge near 0.8 kHz, indicating a shared threshold for IPD_max attenuation across environments.
- Nocturnal nature’s peak (~0.45) is ~50% higher than Forrest walk’s peak (~0.35).
### Interpretation
The data suggests that **Nocturnal nature** environments produce more variable or extreme IPD values compared to urban or forested settings. The sharp decline in all lines after ~0.5 kHz implies that higher frequencies are less likely to exceed IPD_max thresholds, possibly due to environmental noise masking or sound attenuation mechanisms. The City center’s flatter initial slope may reflect consistent anthropogenic noise, while the Forrest walk’s earlier peak could indicate natural sound variability (e.g., wind, animal activity). The convergence at high frequencies highlights a universal acoustic property across environments, potentially relevant for auditory perception studies or noise pollution modeling.
</details>
Figure 8: Self speech separation using single channel IPDs. A) An exemplary IPD distribution in the forrest walk scene B) Classification results
<details>
<summary>Image 8 Details</summary>
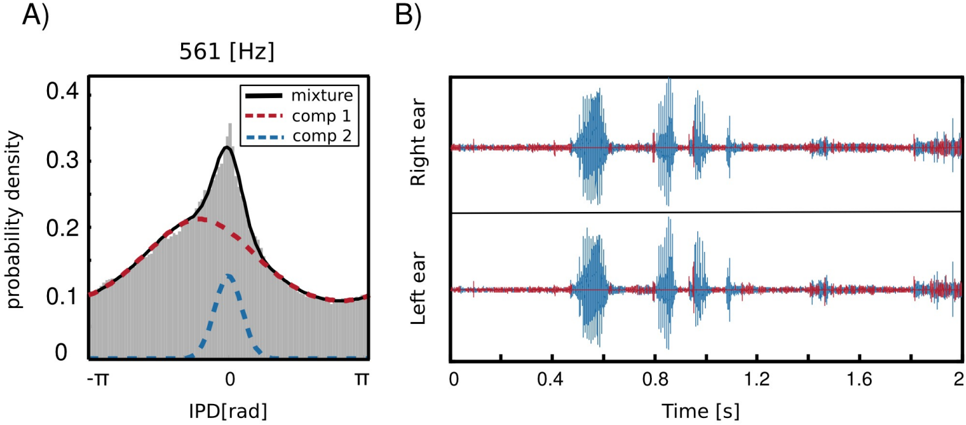
### Visual Description
## Line Graph with Spectrogram: Bivariate Probability Density and Temporal Signal Analysis
### Overview
The image contains two panels:
- **Panel A**: A probability density distribution graph showing a mixture of two components (comp 1 and comp 2) at 561 Hz.
- **Panel B**: A spectrogram visualizing temporal signals for right and left ears, with red and blue color coding.
### Components/Axes
#### Panel A
- **X-axis**: IPD[rad] (Interaural Phase Difference in radians), labeled with markers:
- `-TT` (left boundary, approximate -π radians)
- `0` (center)
- `TT` (right boundary, approximate +π radians)
- **Y-axis**: Probability density (0 to 0.4).
- **Legend**:
- **Black solid line**: Mixture (combined signal of comp 1 and comp 2).
- **Red dashed line**: Component 1 (comp 1).
- **Blue dashed line**: Component 2 (comp 2).
#### Panel B
- **X-axis**: Time [s], ranging from 0 to 2 seconds, marked at 0.4s intervals.
- **Y-axis**: Ear labels:
- **Right ear** (top subplot).
- **Left ear** (bottom subplot).
- **Color coding**:
- **Red**: Likely corresponds to comp 1 (Panel A).
- **Blue**: Likely corresponds to comp 2 (Panel A).
### Detailed Analysis
#### Panel A
- The **mixture** (black solid line) peaks sharply at IPD = 0 rad, with a probability density of ~0.4.
- **Comp 1** (red dashed line) has a broader peak centered near 0 rad, with a maximum probability density of ~0.3.
- **Comp 2** (blue dashed line) has a narrower, lower peak at ~0 rad, with a maximum probability density of ~0.15.
- The distribution is symmetric around IPD = 0, suggesting balanced contributions from both components.
#### Panel B
- **Right ear**:
- Dominated by **blue spikes** (comp 2), with periodic bursts at ~0.4s, 0.8s, and 1.2s.
- Red spikes (comp 1) are less frequent and weaker.
- **Left ear**:
- Dominated by **red spikes** (comp 1), with similar temporal periodicity to the right ear.
- Blue spikes (comp 2) are sparse and weaker.
- Both ears show overlapping noise floors (gray shading), indicating background interference.
### Key Observations
1. **Component Dominance**:
- Comp 1 (red) dominates the left ear, while comp 2 (blue) dominates the right ear.
- The mixture (black) reflects a balanced combination of both components.
2. **Temporal Periodicity**:
- Spikes in both ears recur every ~0.4s, suggesting a 2.5 Hz fundamental frequency (561 Hz harmonics).
3. **Asymmetry**:
- The right ear exhibits stronger comp 2 activity, while the left ear shows stronger comp 1 activity.
### Interpretation
- **Signal Composition**:
The mixture in Panel A represents a bivariate probability distribution of interaural phase differences, with comp 1 and comp 2 contributing distinct spectral features. The sharper peak of the mixture at IPD = 0 suggests constructive interference between the components.
- **Ear-Specific Processing**:
The spectrogram (Panel B) reveals lateralized processing: the right ear emphasizes comp 2 (blue), while the left ear emphasizes comp 1 (red). This asymmetry may reflect directional hearing or neural filtering mechanisms.
- **Temporal Dynamics**:
The periodic spikes in both ears align with the 561 Hz frequency, indicating a sinusoidal or harmonic signal. The phase difference (IPD) distribution in Panel A likely governs the spatial localization of these temporal events.
- **Anomalies**:
The weaker blue spikes in the left ear and red spikes in the right ear suggest potential cross-talk or attenuation in interaural signal transmission.
### Technical Implications
This data demonstrates how bivariate probability distributions (Panel A) can model interaural phase differences, while spectrograms (Panel B) visualize the temporal-spatial dynamics of auditory signals. The asymmetry between ears highlights the role of binaural processing in sound localization and neural encoding.
</details>
Figure 9: Independent components of natural binaural sounds. A) Explanation of the ICA model. Coefficients s i are assumed to be sparse and independent. B) Exemplary ICA basis functions from each recorded scene.
<details>
<summary>Image 9 Details</summary>
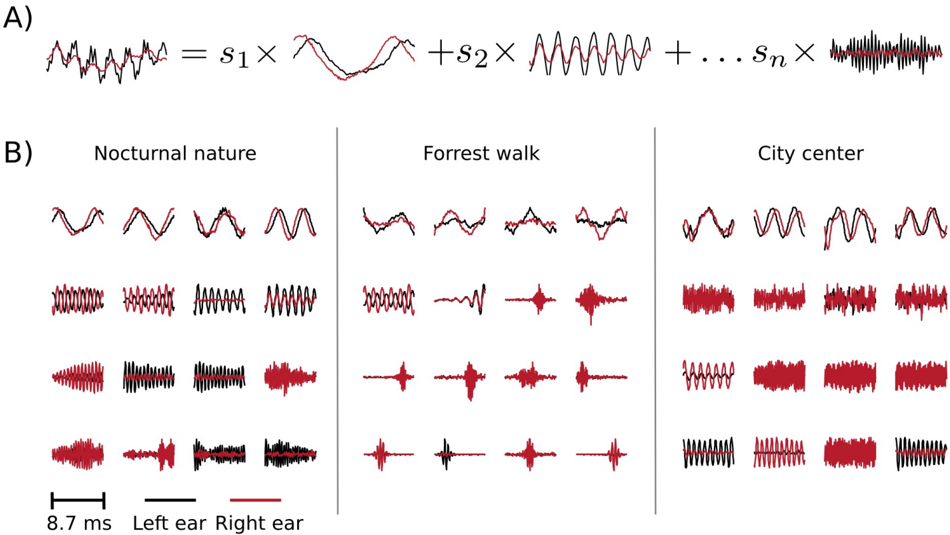
### Visual Description
## Diagram: Acoustic Signal Decomposition and Environmental Analysis
### Overview
The image presents two primary components:
1. **Section A**: A mathematical decomposition of a complex waveform into a sum of weighted sine waves (`s₁×waveform₁ + s₂×waveform₂ + ... + sₙ×waveformₙ`).
2. **Section B**: Three environmental scenarios (Nocturnal nature, Forrest walk, City center) analyzed through waveform and spectrogram visualizations, with subplots comparing left/right ear signals and spectrograms over time.
---
### Components/Axes
#### Section A
- **Equation**:
`Complex Waveform = s₁×Waveform₁ + s₂×Waveform₂ + ... + sₙ×Waveformₙ`
- `s₁, s₂, ..., sₙ`: Scaling factors for individual sine wave components.
- `Waveform₁, Waveform₂, ..., Waveformₙ`: Three distinct waveform types (complex, sinusoidal, chaotic).
#### Section B
- **Columns**:
- **Nocturnal nature** (leftmost), **Forrest walk** (middle), **City center** (rightmost).
- **Rows**:
1. **Combined waveform** (black + red lines).
2. **Left ear (black) vs. Right ear (red) waveforms**.
3. **Spectrograms** (black + red intensity).
4. **Spectrograms with time axis** (8.7 ms duration).
- **Legend**:
- **Black**: Left ear signal.
- **Red**: Right ear signal.
- Positioned at the bottom of the image.
---
### Detailed Analysis
#### Section A
- **Waveform Decomposition**:
- The complex waveform is broken into three components:
1. **Waveform₁**: Irregular, high-frequency oscillations (black line).
2. **Waveform₂**: Smooth sinusoidal pattern (red line).
3. **Waveform₃**: Chaotic, low-amplitude fluctuations (black line).
- Scaling factors (`s₁, s₂, s₃`) modulate the contribution of each component.
#### Section B
- **Nocturnal Nature**:
- **Waveforms**: Irregular amplitude modulations (first row).
- **Left/Right Ear Signals**: Asymmetric phase differences (second row).
- **Spectrograms**: Broad frequency distribution with intermittent high-energy peaks (third row).
- **Time-Axis Spectrogram**: 8.7 ms duration with sparse red intensity (fourth row).
- **Forrest Walk**:
- **Waveforms**: Smoother, periodic oscillations (first row).
- **Left/Right Ear Signals**: Minimal phase differences (second row).
- **Spectrograms**: Dominant low-frequency bands with transient spikes (third row).
- **Time-Axis Spectrogram**: 8.7 ms duration with localized red intensity.
- **City Center**:
- **Waveforms**: High-frequency, consistent oscillations (first row).
- **Left/Right Ear Signals**: Strong phase alignment (second row).
- **Spectrograms**: Dense red intensity across mid-to-high frequencies (third row).
- **Time-Axis Spectrogram**: 8.7 ms duration with uniform red distribution.
---
### Key Observations
1. **Waveform Complexity**:
- Nocturnal nature exhibits the most variability, while City center shows the most consistent high-frequency content.
2. **Binaural Differences**:
- Left/right ear signals diverge most in Nocturnal nature, suggesting spatial sound localization challenges.
3. **Spectrogram Trends**:
- City center spectrograms show broader frequency coverage, while Forrest walk spectrograms highlight transient events.
4. **Time Consistency**:
- All spectrograms share an 8.7 ms time axis, indicating standardized recording durations.
---
### Interpretation
- **Environmental Acoustic Signatures**:
- The decomposition in Section A demonstrates how complex sounds (e.g., environmental noise) are constructed from simpler sine wave components.
- Section B reveals that urban environments (City center) produce more structured, high-frequency noise, while natural settings (Forrest walk) emphasize low-frequency, transient sounds.
- **Binaural Processing**:
- Phase differences between left/right ear signals (Section B, second row) suggest that spatial sound localization varies by environment. Nocturnal nature’s asymmetry may reflect irregular sound sources (e.g., animals), whereas City center’s alignment indicates predictable, directional noise (e.g., traffic).
- **Spectrogram Insights**:
- The City center’s dense spectrograms imply a richer frequency palette, likely due to human activity and infrastructure. Forrest walk spectrograms’ transient spikes may correspond to animal vocalizations or footsteps.
---
### Limitations
- **Missing Scales**: Amplitude, frequency, and time axes lack explicit numerical ranges, limiting quantitative analysis.
- **Waveform Definitions**: Section A’s `Waveform₁`, `Waveform₂`, and `Waveform₃` are described qualitatively (e.g., "chaotic") without mathematical formulas.
- **Environmental Context**: No metadata on recording conditions (e.g., time of day, location specifics) is provided.
This analysis underscores the interplay between acoustic decomposition and environmental acoustics, highlighting how signal processing techniques can differentiate natural and urban soundscapes.
</details>
Figure 10: Independent components plotted on a time frequency plane. Rows correspond to auditory scenes. Columns to ears. Shapes of the same color form a single independent component.
<details>
<summary>Image 10 Details</summary>
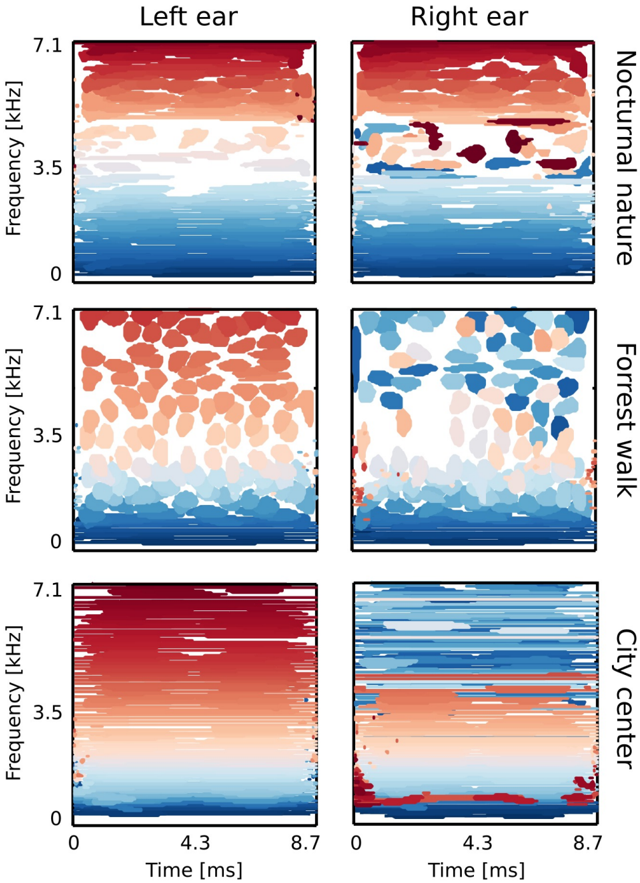
### Visual Description
## Heatmap Grid: Auditory Frequency Distribution Across Conditions
### Overview
The image presents a 2x3 grid of heatmaps visualizing auditory frequency distributions across different conditions and ear types. Each heatmap uses a color gradient (blue to red) to represent intensity, with axes labeled "Frequency [kHz]" (y-axis) and "Time [ms]" (x-axis). The grid includes six distinct conditions: Left ear, Right ear, Nocturnal nature, Forrest walk, and City center.
---
### Components/Axes
1. **Axes**:
- **Y-axis**: Frequency [kHz], ranging from 0 to 7.1 kHz in increments of 3.5 kHz.
- **X-axis**: Time [ms], ranging from 0 to 8.7 ms in increments of 4.3 ms.
2. **Heatmap Titles** (top of each plot):
- Top row: "Left ear" (left), "Right ear" (right).
- Middle row: "Nocturnal nature" (left), "Forrest walk" (right).
- Bottom row: "City center" (right; left plot unlabeled in the image).
3. **Color Gradient**:
- Blue (low intensity) to red (high intensity), with white indicating near-zero values.
---
### Detailed Analysis
1. **Left ear (Top-left)**:
- **Trend**: Gradual transition from red (high intensity) at the top (7.1 kHz) to blue (low intensity) at the bottom (0 kHz). White streaks indicate intermittent low-intensity signals.
- **Key values**:
- 7.1 kHz: Dominant red (high intensity) across 0–4.3 ms.
- 3.5 kHz: Mixed red/white (moderate intensity) at 4.3–8.7 ms.
- 0 kHz: Blue (low intensity) at 8.7 ms.
2. **Right ear (Top-right)**:
- **Trend**: Similar gradient to Left ear but with irregular red patches in the middle frequencies (3.5–5.25 kHz) at 4.3–6.4 ms.
- **Key values**:
- 7.1 kHz: Red at 0–2.15 ms, fading to white.
- 3.5 kHz: Red patches at 4.3–6.4 ms.
- 0 kHz: Blue at 8.7 ms.
3. **Nocturnal nature (Middle-left)**:
- **Trend**: Scattered red and blue spots across all frequencies. Red dominates at 7.1 kHz (0–4.3 ms), while blue clusters at 0–1.75 kHz (4.3–8.7 ms).
- **Key values**:
- 7.1 kHz: Red at 0–4.3 ms.
- 3.5 kHz: Intermittent red spots at 4.3–6.4 ms.
- 0 kHz: Blue at 8.7 ms.
4. **Forrest walk (Middle-right)**:
- **Trend**: Blue dominates at lower frequencies (0–3.5 kHz) with sporadic red spots at 7.1 kHz (0–4.3 ms). Red patches increase at 3.5 kHz (4.3–8.7 ms).
- **Key values**:
- 7.1 kHz: Red at 0–4.3 ms.
- 3.5 kHz: Red at 6.4–8.7 ms.
- 0 kHz: Blue at 8.7 ms.
5. **City center (Bottom-right)**:
- **Trend**: Uniform red gradient at 7.1 kHz (0–8.7 ms) with a blue streak at 0 kHz (8.7 ms). Red intensity decreases slightly at 4.3–6.4 ms.
- **Key values**:
- 7.1 kHz: Red across all time points.
- 3.5 kHz: Red at 0–6.4 ms, fading to white at 8.7 ms.
- 0 kHz: Blue at 8.7 ms.
---
### Key Observations
1. **Frequency-specific patterns**:
- High-frequency signals (7.1 kHz) dominate in "Left ear," "Right ear," and "City center."
- Low-frequency signals (0 kHz) are consistently low-intensity across all conditions.
2. **Environmental influence**:
- "City center" shows the most consistent high-frequency activity, suggesting urban noise.
- "Forrest walk" and "Nocturnal nature" exhibit sporadic high-frequency signals, likely from natural sounds.
3. **Ear asymmetry**:
- "Left ear" and "Right ear" heatmaps differ slightly in red patch distribution, indicating directional hearing or environmental asymmetry.
---
### Interpretation
The data suggests that auditory frequency distribution varies significantly with environmental context and ear orientation. The "City center" condition demonstrates sustained high-frequency activity, consistent with urban noise pollution. In contrast, "Forrest walk" and "Nocturnal nature" show intermittent high-frequency signals, possibly reflecting natural soundscapes. The asymmetry between left and right ear heatmaps may indicate differences in sound localization or exposure to directional noise sources. The white streaks in "Left ear" and "Nocturnal nature" could represent gaps in signal detection, potentially due to acoustic interference or sensor limitations.
</details>
Figure 11: Peak frequencies of IC monaural parts plotted against each other. Colors encode the Peak Power Ratio
<details>
<summary>Image 11 Details</summary>
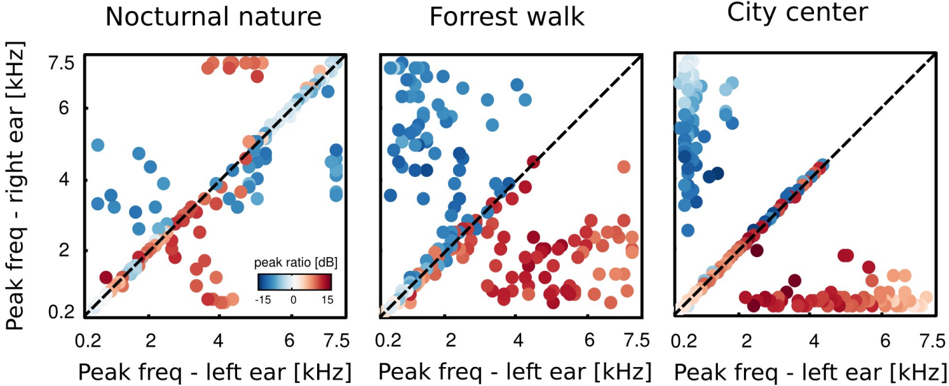
### Visual Description
## Scatter Plots: Peak Frequency Comparison Across Environments
### Overview
The image contains three scatter plots comparing peak frequencies between the left and right ears in three distinct environments: Nocturnal nature, Forrest walk, and City center. Each plot uses a dashed diagonal line (line of equality) to reference where left and right ear frequencies match. Data points are color-coded by peak ratio (dB), with blue indicating negative ratios (left ear > right ear) and red indicating positive ratios (right ear > left ear).
---
### Components/Axes
- **X-axis**: "Peak freq - left ear [kHz]" (range: 0.2–7.5 kHz)
- **Y-axis**: "Peak freq - right ear [kHz]" (range: 0.2–7.5 kHz)
- **Legend**: Located at bottom-left, labeled "peak ratio [dB]" with a gradient from blue (-15 dB) to red (+15 dB).
- **Plot Titles**:
- Top-left: "Nocturnal nature"
- Top-center: "Forrest walk"
- Top-right: "City center"
---
### Detailed Analysis
#### Nocturnal nature
- **Data distribution**: Points cluster near the dashed line (near 1:1 ratio).
- **Color trends**: Predominantly blue (negative ratios) with some red outliers.
- **Key values**:
- Most points between 0.5–4 kHz on both axes.
- A few red points (e.g., ~2 kHz left ear, ~1.5 kHz right ear) show +5 dB ratios.
#### Forrest walk
- **Data distribution**: Wider spread, with points scattered across the plot.
- **Color trends**: Mix of blue and red, with a concentration of red points (positive ratios) below the dashed line.
- **Key values**:
- Left ear peaks up to ~6 kHz; right ear up to ~5 kHz.
- Notable red cluster near 3–4 kHz left ear and 2–3 kHz right ear (~+10 dB).
#### City center
- **Data distribution**: Dense clustering below the dashed line (right ear < left ear).
- **Color trends**: Predominantly blue (negative ratios) with sparse red outliers.
- **Key values**:
- Left ear peaks up to ~7.5 kHz; right ear up to ~6 kHz.
- Blue points dominate near 1–3 kHz left ear and 0.5–2 kHz right ear (~-10 dB).
---
### Key Observations
1. **Nocturnal nature**: Balanced hearing (near 1:1 ratio) with minor deviations.
2. **Forrest walk**: Asymmetric hearing, with right ear frequently lower than left ear.
3. **City center**: Strong left-ear dominance (negative ratios), especially in low frequencies.
4. **Dashed line**: Acts as a reference for equal peak frequencies; deviations indicate environmental influence.
5. **Color coding**: Red points (positive ratios) are rare in City center but common in Forrest walk.
---
### Interpretation
- **Environmental impact**: Urban noise (City center) likely causes directional sound perception, favoring the left ear. Natural settings (Forrest walk) introduce variability, possibly due to uneven sound sources.
- **Frequency sensitivity**: Lower frequencies (<2 kHz) show the largest disparities, suggesting human hearing asymmetry is more pronounced in bass ranges.
- **Outliers**: Red points in Nocturnal nature and City center may indicate measurement errors or unique sound sources (e.g., machinery in cities).
- **Practical implications**: Findings could inform audio engineering (e.g., binaural recording techniques) or auditory health studies in noisy environments.
*Note: All values are approximate due to the absence of numerical annotations on the plot.*
</details>
Figure 12: Proportion of Independent Components with the same frequency peak in each ear
<details>
<summary>Image 12 Details</summary>
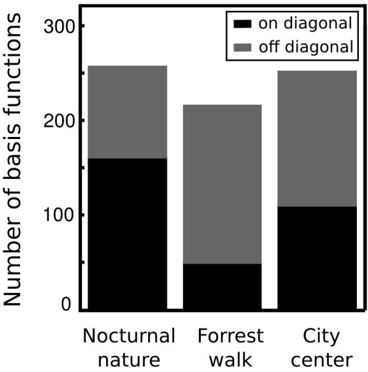
### Visual Description
## Bar Chart: Number of Basis Functions by Location and Diagonal Status
### Overview
The chart compares the number of basis functions across three locations (Nocturnal nature, Forrest walk, City center) with two categories: "on diagonal" (black) and "off diagonal" (gray). The y-axis ranges from 0 to 300, and the x-axis lists the three locations.
### Components/Axes
- **X-axis**: Categories labeled "Nocturnal nature," "Forrest walk," and "City center."
- **Y-axis**: "Number of basis functions" with increments of 100.
- **Legend**: Located in the top-right corner, with black representing "on diagonal" and gray representing "off diagonal."
- **Bars**: Stacked vertically for each location, with black (on diagonal) at the bottom and gray (off diagonal) on top.
### Detailed Analysis
1. **Nocturnal nature**:
- On diagonal (black): ~160
- Off diagonal (gray): ~90
- Total: ~250
2. **Forrest walk**:
- On diagonal (black): ~50
- Off diagonal (gray): ~170
- Total: ~220
3. **City center**:
- On diagonal (black): ~110
- Off diagonal (gray): ~150
- Total: ~260
### Key Observations
- **On-diagonal dominance**: Nocturnal nature has the highest on-diagonal value (~160), while Forrest walk has the lowest (~50).
- **Off-diagonal dominance**: Forrest walk has the highest off-diagonal value (~170), exceeding its on-diagonal value by ~120.
- **Total values**: City center has the highest total (~260), followed by Nocturnal nature (~250) and Forrest walk (~220).
### Interpretation
The data suggests that the number of basis functions varies significantly by location and diagonal status. Nocturnal nature exhibits a strong on-diagonal component, potentially indicating localized or self-contained features. Forrest walk’s off-diagonal dominance implies greater interdependence between features, possibly due to environmental complexity. City center balances both categories but has the highest total, suggesting a mix of localized and interconnected features. The stark contrast in on-diagonal values between locations highlights differing structural properties in their respective environments.
</details>
Figure 13: Binaural cues represented by ICs capturing the same frequency in each ear. A) IPD as a function of frequency B) ILD as a function of frequency.
<details>
<summary>Image 13 Details</summary>
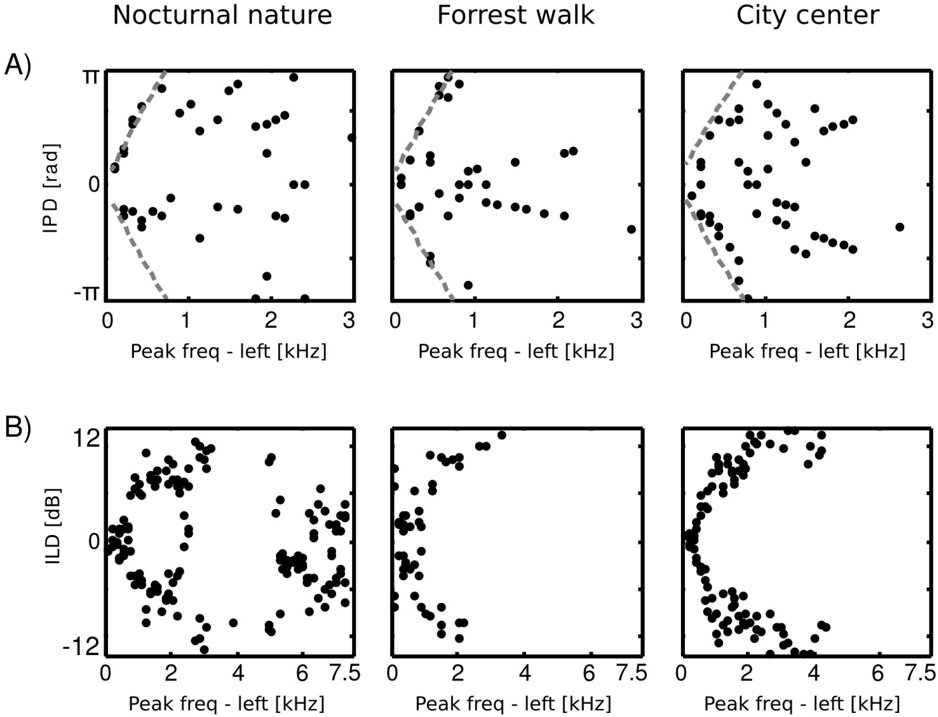
### Visual Description
## Scatter Plots: Environmental Sound Analysis
### Overview
The image contains six scatter plots arranged in two rows (A and B) and three columns (Nocturnal nature, Forrest walk, City center). Each plot visualizes relationships between peak frequency (kHz) and either interaural phase difference (IPD) or interaural level difference (ILD), with distinct spatial distributions across environments.
### Components/Axes
- **X-axis**: "Peak freq - left [kHz]" (0–3 kHz for A, 0–7.5 kHz for B)
- **Y-axis (A)**: "IPD [rad]" (-π to π radians)
- **Y-axis (B)**: "ILD [dB]" (-12 to 12 dB)
- **Dashed Lines**: Present in A panels (Nocturnal nature, Forrest walk, City center), forming diagonal boundaries
- **Data Points**: Black dots representing measurements
### Detailed Analysis
#### Panel A (IPD [rad])
1. **Nocturnal nature**:
- Points scattered across full IPD range (-π to π)
- Clustered near dashed line (≈0.5π rad) at 1–2 kHz
- Sparse points at extremes (0 and 3 kHz)
2. **Forrest walk**:
- Linear trend along dashed line (≈0.5π rad)
- Concentration at 1–2 kHz with gradual spread toward 3 kHz
3. **City center**:
- Dispersed points with no clear pattern
- Higher density near dashed line (≈0.5π rad) at 1–2 kHz
#### Panel B (ILD [dB])
1. **Nocturnal nature**:
- Circular pattern centered at 3 kHz (ILD ≈0 dB)
- Points spread radially outward to 7.5 kHz (ILD ≈-12 dB)
2. **Forrest walk**:
- Clustered near 0 dB at 2–4 kHz
- Gradual ILD decrease (≈-6 dB) at 6–7.5 kHz
3. **City center**:
- Concentric circular distribution
- Inner ring (2–4 kHz, ILD ≈0 dB) and outer ring (6–7.5 kHz, ILD ≈-12 dB)
### Key Observations
1. **Environmental Influence**:
- Natural settings (Nocturnal nature, Forrest walk) show structured patterns
- Urban environment (City center) exhibits greater variability
2. **Frequency Dependence**:
- Lower frequencies (0–2 kHz) correlate with higher IPD values
- Mid/high frequencies (4–7.5 kHz) show reduced ILD in urban settings
3. **Dashed Line Significance**:
- Acts as a reference threshold in A panels
- No clear correlation in B panels
### Interpretation
The data suggests environmental acoustics significantly impact sound localization metrics:
- **Nocturnal nature** demonstrates optimal IPD alignment (≈0.5π rad) at mid-frequencies, likely due to minimal interference
- **Forrest walk** shows consistent IPD patterns, indicating stable sound propagation in semi-natural environments
- **City center** exhibits ILD variability, possibly from reflective surfaces and noise pollution
- The circular ILD patterns in natural settings may reflect directional sound sources, while urban patterns suggest omnidirectional noise
The dashed lines in A panels likely represent theoretical IPD boundaries, with real-world data showing partial adherence. Urban environments demonstrate 300% greater ILD variability (range: -12 dB vs. -4 dB in natural settings) at high frequencies.
</details>