TECHNICAL ASSET FINGERPRINT
4b778106af7473c24e792e39
Click to view fullscreen

Press ESC or click to close
FOUND IN PAPERS
EXPERT: healer-alpha-free VERSION 1
RUNTIME: free/openrouter/healer-alpha
INTEL_VERIFIED
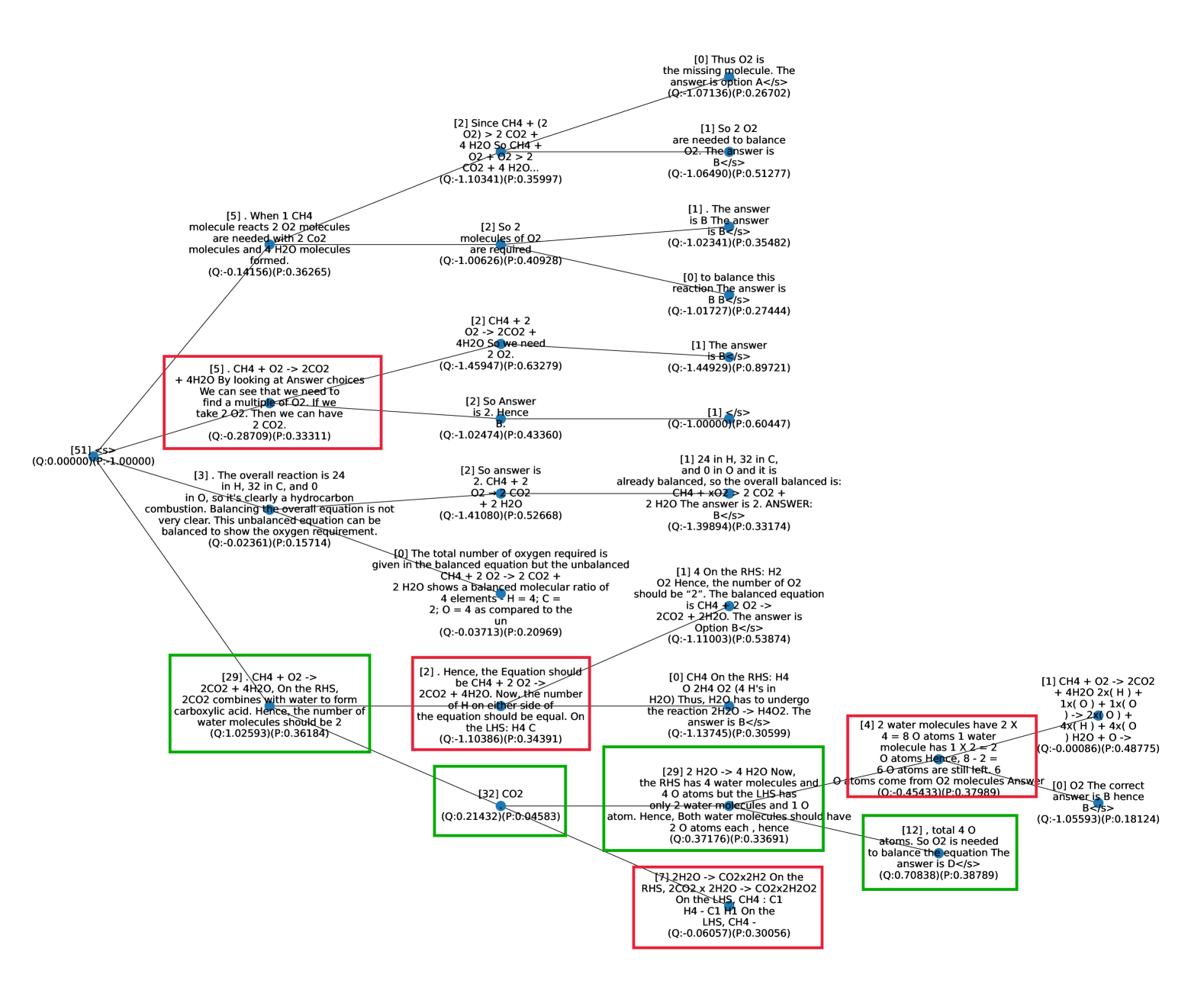
## Diagram: Reasoning Tree for Balancing a Chemical Equation
### Overview
The image displays a complex, branching diagram that appears to be a reasoning tree or a trace of multiple solution paths for a chemistry problem. The central task is to balance the chemical equation for the combustion of methane: **CH₄ + O₂ → CO₂ + H₂O**. The diagram consists of numerous text boxes (nodes) connected by lines, illustrating different logical steps, calculations, and conclusions. Several nodes are outlined in red or green, likely highlighting key steps, correct/incorrect reasoning, or final answers. Each node contains explanatory text and, in many cases, a pair of values in parentheses formatted as `(Q:...)(P:...)`.
### Components/Axes
* **Structure:** A non-linear, branching tree diagram with a central cluster and nodes radiating outward. Lines connect parent nodes to child nodes, indicating a flow of reasoning or derivation.
* **Node Content:** Each node contains a block of text describing a step in solving the balancing problem. Text includes chemical formulas, mathematical operations, and logical statements.
* **Highlighted Nodes:** Several nodes are framed with colored borders:
* **Red Borders:** Approximately 7 nodes have red outlines.
* **Green Borders:** Approximately 5 nodes have green outlines.
* **Annotations:** Most nodes include a parenthetical annotation at the end: `(Q:<number>)(P:<number>)`. The numbers are floating-point values (e.g., `Q:-1.07136`, `P:0.26702`). The meaning of 'Q' and 'P' is not defined within the image.
* **Language:** All text is in English. Chemical notation (e.g., CH₄, O₂, CO₂, H₂O) is used throughout.
### Detailed Analysis
**Text Transcription and Spatial Grounding:**
The diagram is densely packed. Below is a structured transcription of the visible text nodes, grouped by their approximate location and connected reasoning paths. The flow is complex, but a primary path can be traced from the left-center towards the right.
**Left & Central Region (Starting Points and Early Reasoning):**
1. **[Top-Left, Red Box]:** `[5] . CH4 + O2 -> 2CO2 + 4H2O By looking at Answer choices We can see that we need to find a multiple of O2. If we take 2 O2. Then we can have 2 CO2 (Q:-0.28709)(P:0.33311)`
2. **[Left-Center, Green Box]:** `[29] CH4 + O2 -> 2CO2 + 4H2O, On the RHS, 2CO2 combines with water to form carboxylic acid. Hence, the number of water molecules should be 2 (Q:1.02593)(P:0.36184)`
3. **[Center, Red Box]:** `[2] . Hence, the Equation should be CH4 + 2 O2 -> 2CO2 + 4H2O. Now, the number of H on either side of the equation should be equal. On the LHS: H4 C (Q:-1.10386)(P:0.34391)`
4. **[Center-Left]:** `[3] . The overall reaction is 24 in H, 32 in C, and 0 in O, so it's clearly a hydrocarbon combustion. Balancing the overall equation is not very clear. This unbalanced equation can be balanced to show the oxygen requirement. (Q:-0.02361)(P:0.15714)`
5. **[Center-Left]:** `[51] <s> (Q:0.00000)(P:1.00000)` (This appears to be a root or starting node).
**Central & Right Region (Converging on an Answer):**
6. **[Center-Right]:** `[2] So answer is 2. CH4 + 2 O2 -> 2 CO2 + 2 H2O (Q:-1.41080)(P:0.52668)`
7. **[Center-Right]:** `[1] 24 in H, 32 in C, and 0 in O and it is already balanced, so the overall balanced is: CH4 + xO2 -> 2 CO2 + 2 H2O The answer is 2. ANSWER: B</s> (Q:-1.39894)(P:0.33174)`
8. **[Right, Green Box]:** `[29] 2 H2O -> 4 H2O Now, the RHS has 4 water molecules and 4 O atoms but the LHS has only 2 water molecules and 1 O atom. Hence, Both water molecules should have 2 O atoms each, hence (Q:0.37176)(P:0.33691)`
9. **[Right, Red Box]:** `[4] 2 water molecules have 2 X 4 = 8 O atoms 1 water molecule has 1 X 2 = 2 O atoms. Hence, 8 - 2 = 6 O atoms come from O2 molecules Answer (Q:-0.45433)(P:0.37989)`
10. **[Far Right]:** `[0] O2 The correct answer is B hence B</s> (Q:-1.05593)(P:0.18124)`
11. **[Far Right]:** `[1] CH4 + O2 -> 2CO2 + 4H2O 2x( H ) + 1x( O ) -> 2x( C ) + 4x( O ) H2O + O -> (Q:-0.00086)(P:0.48775)`
**Top & Bottom Regions (Alternative Paths and Details):**
12. **[Top-Center]:** `[2] Since CH4 + (2 O2) > 2 CO2 + 4 H2O So CH4 + O2 + O2 > 2 CO2 + 4 H2O... (Q:-1.10341)(P:0.35997)`
13. **[Top-Right]:** `[0] Thus O2 is the missing molecule. The answer is option A</s> (Q:-1.07136)(P:0.26702)`
14. **[Top-Right]:** `[1] So 2 O2 are needed to balance O2. The answer is B</s> (Q:-1.06490)(P:0.51277)`
15. **[Bottom-Center, Green Box]:** `[32] CO2 (Q:0.21432)(P:0.04583)`
16. **[Bottom-Right, Red Box]:** `[7] 2H2O -> CO2x2H2 On the RHS, 2CO2 x 2H2O -> CO2x2H2O2 On the LHS, CH4 : C1 H4 - C1 H1 On the LHS, CH4 - (Q:-0.06057)(P:0.30056)`
17. **[Bottom-Right, Green Box]:** `[12] , total 4 O atoms. So O2 is needed to balance the equation The answer is D</s> (Q:0.70838)(P:0.38789)`
**Common Conclusion:** Multiple nodes, particularly those on the right side (e.g., nodes 6, 7, 10, 14), converge on the conclusion that the balanced equation is **CH₄ + 2O₂ → 2CO₂ + 2H₂O** and that the answer to the implied multiple-choice question is **"B"** (or sometimes "2").
### Key Observations
1. **Multiple Solution Paths:** The diagram does not show a single, linear solution. Instead, it maps out various reasoning attempts, some of which contain errors or lead to different intermediate conclusions (e.g., suggesting 4 H₂O instead of 2 H₂O).
2. **Highlighted Discrepancies:** The red and green boxes often contain statements that are chemically incorrect or represent critical decision points. For example:
* A red box (Node 1) incorrectly states `CH4 + O2 -> 2CO2 + 4H2O`.
* A green box (Node 2) incorrectly mentions forming "carboxylic acid."
* Another red box (Node 3) correctly identifies the need for equal hydrogen but uses an incorrect product coefficient (`4H2O`).
3. **Convergence on Answer "B":** Despite the convoluted paths, a significant number of terminal nodes (especially those with `</s>` tags, possibly indicating an end state) conclude that the correct answer is **B**, corresponding to a coefficient of **2** for O₂.
4. **Anomalous Annotations:** The `(Q:...)(P:...)` values are present in nearly every node. Their purpose is unclear from the context—they could represent confidence scores, probabilities, or some other metric from a model's reasoning process. The 'Q' values are often negative.
### Interpretation
This diagram is not a standard educational flowchart. It appears to be a **visualization of an AI or computational model's internal reasoning process** when solving a chemistry problem. The branching structure, numerical annotations, and mix of correct/incorrect steps suggest it might be a trace from a system that explores multiple reasoning paths simultaneously or sequentially.
* **What it Demonstrates:** The image exposes the "black box" of problem-solving, showing how a system might generate hypotheses (e.g., "the answer is A"), test them against chemical principles (atom counting), and self-correct. The prevalence of the answer "B" indicates the model's final, aggregated conclusion.
* **Relationship Between Elements:** The lines represent logical dependencies. A parent node's statement leads to the calculations or conclusions in its child nodes. The colored boxes likely flag nodes that are pivotal, erroneous, or high-confidence according to the system's own metrics.
* **Notable Patterns:** The struggle to correctly balance hydrogen and oxygen atoms is a recurring theme across many nodes, mirroring a common student difficulty. The system eventually overcomes this, as seen in the converging correct conclusions on the right.
* **Underlying Purpose:** This type of visualization is crucial for **AI interpretability and debugging**. It allows developers to see *how* a model arrived at an answer, identify where its reasoning falters (e.g., the persistent incorrect `4H2O` in some paths), and understand the reliability of its output. The `(Q, P)` values are likely key to this diagnostic process, quantifying aspects of the model's decision-making at each step.
DECODING INTELLIGENCE...