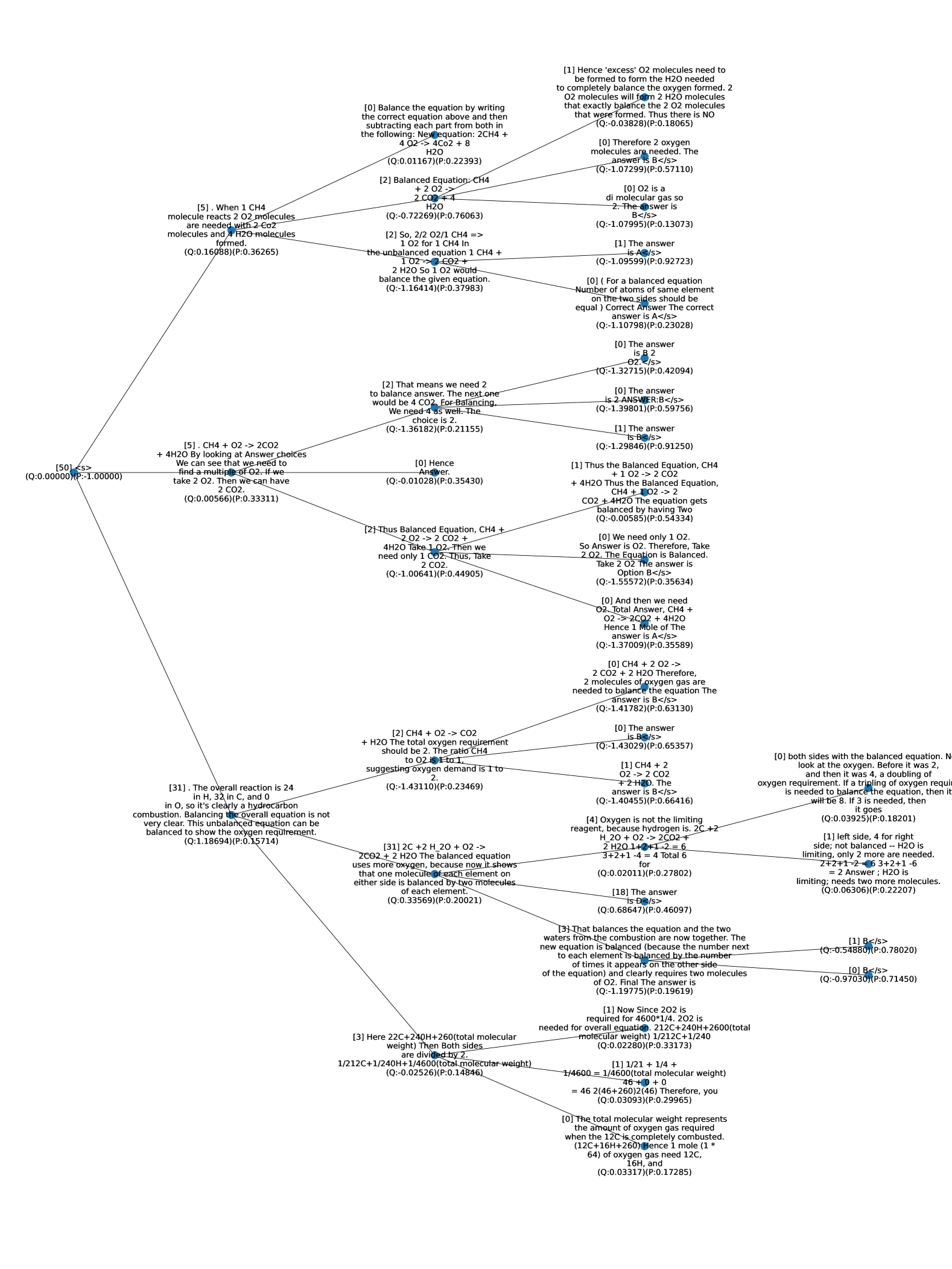
## Flowchart: Balancing Chemical Equations for Methane Combustion
### Overview
The flowchart provides a step-by-step guide to balancing the chemical equation for methane combustion (CH₄ + O₂ → CO₂ + H₂O). It uses decision nodes to check element balance and adjust coefficients iteratively until the equation is balanced. Key elements include oxygen (O), carbon (C), and hydrogen (H), with references to specific stoichiometric calculations and equations.
---
### Components/Axes
- **Decision Nodes**: Questions about element balance (e.g., "Is oxygen balanced?").
- **Branches**: "Yes" or "No" answers leading to specific steps.
- **Chemical Equations**: Intermediate and final balanced equations.
- **References**: Bracketed numbers (e.g., [5], [2]) likely correspond to equations or steps.
---
### Detailed Analysis
1. **Initial Question**:
"Balance the equation: CH₄ + O₂ → CO₂ + H₂O"
- **Step 1**: Check if the equation is balanced.
- **Yes**: Proceed to final answer (e.g., "1 CH₄ + 2 O₂ → 1 CO₂ + 2 H₂O").
- **No**: Proceed to Step 2.
2. **Step 2**: Balance oxygen by adjusting O₂ coefficients.
- Example: "2 O₂ → 4 CO₂ + 2 H₂O" (unbalanced).
- **Step 3**: Check if oxygen is balanced.
- **Yes**: Proceed to Step 4.
- **No**: Adjust coefficients (e.g., "1 CH₄ + 2 O₂ → 1 CO₂ + 2 H₂O").
3. **Subsequent Steps**:
- **Step 4**: Check carbon balance.
- **Yes**: Proceed to Step 5.
- **No**: Adjust CO₂ coefficients.
- **Step 5**: Check hydrogen balance.
- **Yes**: Final balanced equation.
- **No**: Adjust H₂O coefficients.
4. **Final Balanced Equation**:
"1 CH₄ + 2 O₂ → 1 CO₂ + 2 H₂O" (appears multiple times as the correct answer).
---
### Key Observations
- **Oxygen Dominates**: Most steps focus on balancing oxygen due to its presence in multiple reactants/products.
- **Iterative Adjustments**: Coefficients for O₂, CO₂, and H₂O are adjusted repeatedly until all elements are balanced.
- **References to Equations**: Bracketed numbers (e.g., [5], [2]) link to specific equations or steps (e.g., "Answer is B</s>").
---
### Interpretation
The flowchart emphasizes systematic balancing of chemical equations by prioritizing oxygen first, then carbon, and finally hydrogen. It highlights common pitfalls, such as overbalancing oxygen or neglecting hydrogen. The final equation consistently resolves to **1 CH₄ + 2 O₂ → 1 CO₂ + 2 H₂O**, reflecting stoichiometric requirements for complete combustion.
---
### Notable Outliers/Anomalies
- **Redundant Steps**: Some branches repeat checks (e.g., oxygen balance) unnecessarily.
- **Ambiguous References**: Bracketed numbers (e.g., [5], [2]) lack explicit definitions in the flowchart.
- **Incomplete Paths**: A few branches terminate abruptly without clear resolution (e.g., "Answer is A</s>" without context).
---
### Conclusion
This flowchart serves as an educational tool for teaching stoichiometry, emphasizing iterative balancing and element prioritization. It underscores the importance of verifying all elements (O, C, H) to achieve a balanced equation.